Thyroid dysfunction in individuals visiting a Clinic in Lalitpur, Nepal
Rajendra Maharjan,1,2 Ashish Rouniyar1,3*
¹Department of Clinical Laboratories, Genesis Diagnostic and Clinic, Lalitpur, Nepal
²Department of Pathology, Nepal Armed Police Force Hospital, Kathmandu, Nepal
³Department of Microbiology, Nepal Police Hospital, Kathmandu, Nepal
*Corresponding author
*Aashish Gupta, Department of Microbiology, Nepal Police Hospital, Kathmandu, Nepal.
DOI: 10.55920/JCRMHS.2025.09.001385
NTIS= non-thyroidal illness syndrome, HTT=hypertriiodothyroninemia, EH=euthyroid hyperthyroxinemia
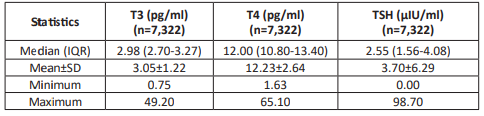
Table 2: Descriptive statistics of serum thyroid marker levels analyzed among clinic-visiting individuals
Key Findings by Disorder Type
Hyperthyroidism was rare in this cohort, with a specific distribution among primary hyperthyroidism patients in the 20-29 and 30-39 age groups, where they form 44.44% and 55.56% of cases, respectively. Hypothyroidism cases were primarily clustered in the subclinical and primary forms, with nearly 72% of the total hypothyroidism cases being subclinical. Non-thyroidal illness syndrome, found exclusively in males, primarily affects those over 50, suggesting that non-thyroidal illness syndrome may correlate with older male patients, possibly influenced by comorbidities. Lastly, hypertriiodothyroninemia, although uncommon, is entirely male and has notable distribution in the 10-19 age group, pointing toward a possible age and gender predisposition for hypertriiodothyroninemia (Table 1).
Descriptive statistics of thyroid markers
A total of 7,322 individuals visiting the clinic were analyzed for thyroid function, as measured by serum levels of T3, T4, and TSH. The median T3 concentration was 2.98 pg/ml with an interquartile range (IQR) of 2.70 to 3.27 pg/ml, while the mean±SD was 3.05±1.22 pg/ml. For T4, the median was 12.00 pg/ml (IQR: 10.80–13.40 pg/ml), and the mean±SD was 12.23±2.64 pg/ml. The median TSH level was 2.55 µIU/ml (IQR: 1.56–4.08 µIU/ml), with a mean±SD of 3.70±6.29 µIU/ml (Table 2).
FT3=free triiodothyronine, FT4=free thyroxine, TSH=thyroid stimulating hormone, IQR= interquartile range, SD=standard deviation
Table 3: Statistical assessments of thyroid markers on healthy individuals and patients with thyroid disorders
FT3=free triiodothyronine, FT4=free thyroxine, TSH=thyroid stimulating hormone p-values for patients with thyroid dysfunction were calculated using an independent-sample t-test, with healthy individuals serving as the comparison group
Figure 1: Assessment of normal distribution of thyroid markers
FT3=free triiodothyronine, FT4=free thyroxine, TSH=thyroid stimulating hormone
T3=triiodothyronine, T4=thyroxine, TSH=thyroid stimulating hormone, dotted-black line represents median value of TSH, dotted-red line represents median value of FT4, dotted-blue line represents median value of FT3, shaded region represents values in normal range
Figure 2: Scatter plots exhibiting correlations among thyroid biomarkers
Comparative analysis between healthy individuals and thyroid dysfunction cases
Using an independent-sample t-test, thyroid marker levels were compared between healthy individuals and patients diagnosed with various forms of thyroid dysfunction. The mean±SD levels of FT3, FT4, and TSH in healthy individuals were 2.99±0.40 pg/ml, 12.22±1.69 pg/ml, and 2.26±1.06 µIU/ml, respectively. In cases of patients with primary hyperthyroidism (n=30), significantly elevated FT3 (10.72±11.18 pg/ml, p=0.001) and FT4 (26.82±11.32 pg/ml, p<0.001) levels, along with markedly low TSH (0.01±0.01 µIU/ml, p<0.001), were observed. Patients with secondary hyperthyroidism (n=9) also showed high FT3 (5.81±1.50 pg/ml, p<0.001) and FT4 (20.66±2.62 pg/ml, p<0.001) levels; however, TSH levels (14.46±19.98 µIU/ml) did not reach statistical significance (p=0.104). In subclinical hyperthyroidism (n=101), TSH was significantly reduced (1.32±1.33 µIU/ml, p<0.001), with slightly elevated FT3 and FT4 levels (p<0.001) (Table 3).
group.
Thyroid marker levels in hypothyroidism and other conditions
For patients with primary hypothyroidism (n=164), FT3 and FT4 levels were significantly reduced (2.76±0.80 pg/ml, p<0.001 and 10.38±6.18 pg/ml, p<0.001, respectively), whereas TSH was substantially elevated (22.07±25.31 µIU/ml, p<0.001). Patients with secondary hypothyroidism (n=19) similarly exhibited lower FT3 (1.53±0.40 pg/ml) and FT4 (6.65±2.21 pg/ml) levels, with TSH levels also diminished (1.22±1.18 µIU/ml, p<0.001). Patients with subclinical hypothyroidism (n=1,309) exhibited normal FT3 levels (3.00±0.52 pg/ml, p=0.688), with a moderate reduction in TSH (7.92±7.95 µIU/ml, p<0.001), suggesting a distinct pattern compared to overt hypothyroidism (Table 3).
Thyroid marker abnormalities in patients with other thyroidal illnesses
Patients with euthyroid hyperthyroxinemia (n=25) had elevated FT3 (7.31±6.26 pg/ml, p=0.02) and FT4 (23.15±5.08 pg/ml, p<0.001) levels but normal TSH levels (2.38±1.35 µIU/ml, p=0.578). In patients with hypertriiodothyroninemia (n=148), FT3 was slightly elevated (3.75±4.12 pg/ml, p=0.028), whereas FT4 and TSH levels remained within the normal range. Patients with non-thyroidal illness syndrome (n=104) had significant reductions in FT4 (8.13±1.15 pg/ml, p<0.001) and slight reductions in TSH (2.58±1.10 µIU/ml, p=0.002), while FT3 remained within normal limits (p<0.001) (Table 3).
Distribution of serum level of thyroid biomarkers
The serum levels of T3 (Kolmogorov-Smirnov; p<0.001), T4 (Kolmogorov-Smirnov; p<0.001), and TSH (Kolmogorov-Smirnov; p<0.001) in clinic visitors were not normally distributed (Figure 1).
Correlation analysis of thyroid markers
Figure 2 presents scatter plots depicting the relationships among serum levels of T3, T4, and TSH in clinic visitors. The T3 versus TSH plot shows diffuse clustering with no statistically significant correlation (r=-0.022, p=0.060), indicating minimal association between these markers. Conversely, the T4 versus TSH plot displays a weak but significant inverse correlation (r=-0.123, p<0.001), suggesting that higher FT4 levels are associated with lower TSH levels (Figure 2).