Hemolytic Anemia Due to Severe Vitamin B12 Deficiency Associated with Pernicious Anemia: A Case Report
Mustafa Şentürk1*, Alara Tavares2, Deniz Arıca3, Aylin Yıldırır4
¹Department of Internal Medicine, Başkent University Faculty of Medicine, Ankara, Turkey.
²Medicine Student, Başkent University Faculty of Medicine, Ankara, Turkey.
³Department of Medical Hematology, Başkent University Faculty of Medicine, Ankara, Turkey.
⁴Department of Medical Cardiology, Başkent University Faculty of Medicine, Ankara, Turkey.
*Corresponding author
*Dr. Mustafa Şentürk, Department of Internal Medicine, Başkent University Faculty of Medicine, Ankara, Turkey.
Email: senturk96mustafa@hotmail.com
DOI: 10.55920/JCRMHS.2024.08.001348
Figure 1: The patient's peripheral blood smear. A: Hypersegmented neutrophil. B: Macrocytic erythrocyte.
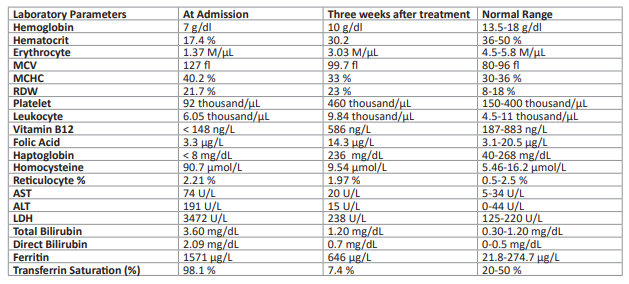
Table 1: Laboratory results at the time of admission and three weeks after treatment.
Transthoracic echocardiography revealed moderate degree aortic stenosis (aortic mean gradient 27mmHg, mean aort valve are of 1.0 cm2) and severely depressed left ventricular systolic function (left ventricular ejection fraction of 32%). A transesophageal echocardiogram (TEE) was performed after atrial fibrillation was observed on the follow-up ECG of the patient, whose initial ECG showed sinus rhythm. TEE showed dense spontaneous echo contrast (SEC) in the left atrium, with a 1.2x0.7 cm thrombus in the left atrial appendage. Therefore, cardioversion could not be performed, and the patient was planned to be monitored with anticoagulation therapy. Esophagogastroduodenoscopy, planned due to anti-parietal antibody positivity, was postponed after cardiac further evaluations of the patient. The patient was discharged with a prescription for apixaban 2x2.5 mg and scheduled for follow-up in 3 weeks. Cardiac catheterization and coronary angiography were planned to investigate the etiology of left ventricular dysfunction after correction of the anemia.
Three weeks after treatment, laboratory tests showed improvement in bicytopenia with a hemoglobin level of 10 g/dL, platelets of 460000/μL, and leukocytes of 9,840/μL. The MCV value was 99.7 fl, haptoglobin was 236 mg/dL, and reticulocyte count was 1.97%. Total bilirubin decreased to 1.20 mg/dL, ALT to 20 U/L, AST to 15 U/L, and LDH to 238 U/L. Regarding other nutritional parameters, ferritin was 646 μg/L and transferrin saturation was 7.4%. The patient's vitamin B12 level was 586 ng/L, homocysteine was 9.54 μmol/L, and folic acid level was 14.3 μg/L. The patient's complaints of fatigue, tachycardia, and shortness of breath completely resolved, and a follow-up appointment was scheduled for one month later.