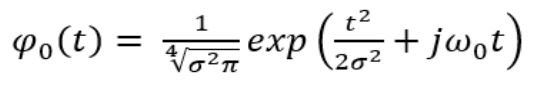Tracking the Recovery of a Mild Traumatic Brain Injury Patient utilizing a 60-s Combined Functional Near-Infrared Spectroscopy and Psychomotor Testing Approach: A Case Study
Cory M. Smith*, Cierra B. Ugale, Matt D. Segovia, Katie M. Lee, Andrew R. Gallucci, Joshua R. Thompson, Hunter D. Dobbs, Owen F. Salmon
Robbins College of Health and Human Sciences, Human & Environmental Physiology Laboratory, Baylor University, Waco, TX USA.
*Corresponding author
*Dr. Cory M. Smith, Assistant Professor Human & Environmental Physiology Laboratory Baylor University One Bear Place #97313 Waco, Texas 76798, USA.
DOI: 10.55920/JCRMHS.2024.07.001279
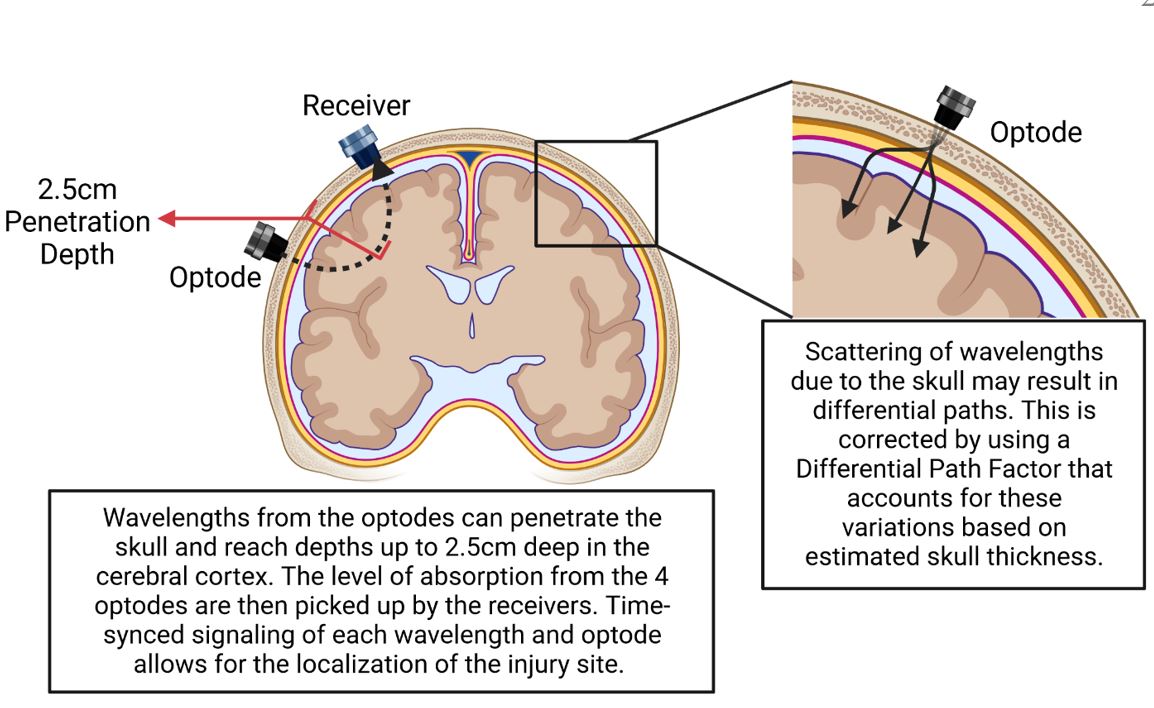
Figure 1: Functional Near-Infrared Spectroscopy (fNIRS) setup for the impacted and unimpacted region of the head in the patient. This displays the 4 optode and receiver placements 4 x 1 grid approach utilized in this study. The optodes cycled 2 wavelengths of light, each at 10Hz to be detected with the receiver located in the center of the 4x1 cluster.
Equation (3) μa (λ)-εH2O (λ) cH2O=εO2Hb (λ)cO2Hb+εHHbHHb (λ)cHHb
Equation 3 is involved in the 1st order processing and corrects for the water content within the signal absorption rates. ε = extinction coefficient, λ = wavelength, c = concentration
Equation (4)
Equation 4 provides the individual channel calculation in each prefrontal cortex hemisphere Morlet wavelet analysis. The CWT transform will be utilized with a 20 scale and 512 time-step coefficients without any overlap. The Wavelet coefficients were determined from the culmination of all the CWT data over each individual epoch to determine the Morlet CWT frequency of the signal. The data from the Morlet Wavelet was further analyzed with CWT Multiscale Peak Detection to quantify the amplitude of each channel, hemisphere, and timepoint. This analysis will allow for the calculation of fNIRSamp values for O2Hb, HHb. And tHb. Each hemispheres 4 optode grid channels relative measurements was then summated to provide an singular value for each hemisphere averaged across the 60-s PVT for each of the 10 weeks.
Three exploratory t-tests were performed on the mean Control and Injured tHbamp, HHbamp, and O2Hbamp measures, collapsed across the 10 weeks for sufficient data points, to determine the gross differences in hemodynamics throughout the 60-s PVT.
Psychomotor Vigilance Test
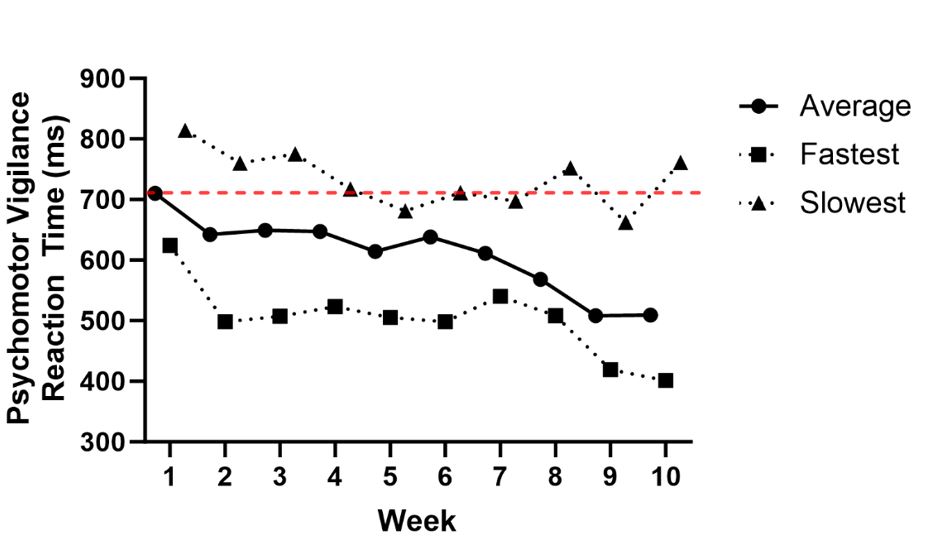
A 60-s PVT was performed each week while wearing the fNIRS neuroimaging sensor on the injured and control regions of the patient’s brain. The 60-s PVT test was performed on a touch screen tablet (iPad 10.2in 9th generation, Apple, Cupertino, CA) using the Research Buddies software (Research Buddies Version 1.53). During each visit, the patients’ index finger on the dominant hand was placed on the lower corner of the tablet. The researcher then started the 60-s PVT, and the patient would tap the center of the screen as quickly as possible when a number appeared. Upon completion of the 60-s PVT, the average, fastest, and slowest reaction time were recorded (Figure 3).
Figure 2: Visualization of the fundamental principles underlying functional near-infrared spectroscopy (fNIRS) for cerebral hemodynamics from the perspective of one of the 4 optodes utilized in each 4x1 grid.
Figure 3: Results of the 60-s Psychomotor Vigilance Reaction Time test including the average, fastest, and slowest reaction time for each weekly visit during the 10-wk recovery period. The dashed line represents the average reaction time captured within 72-hr of injury.
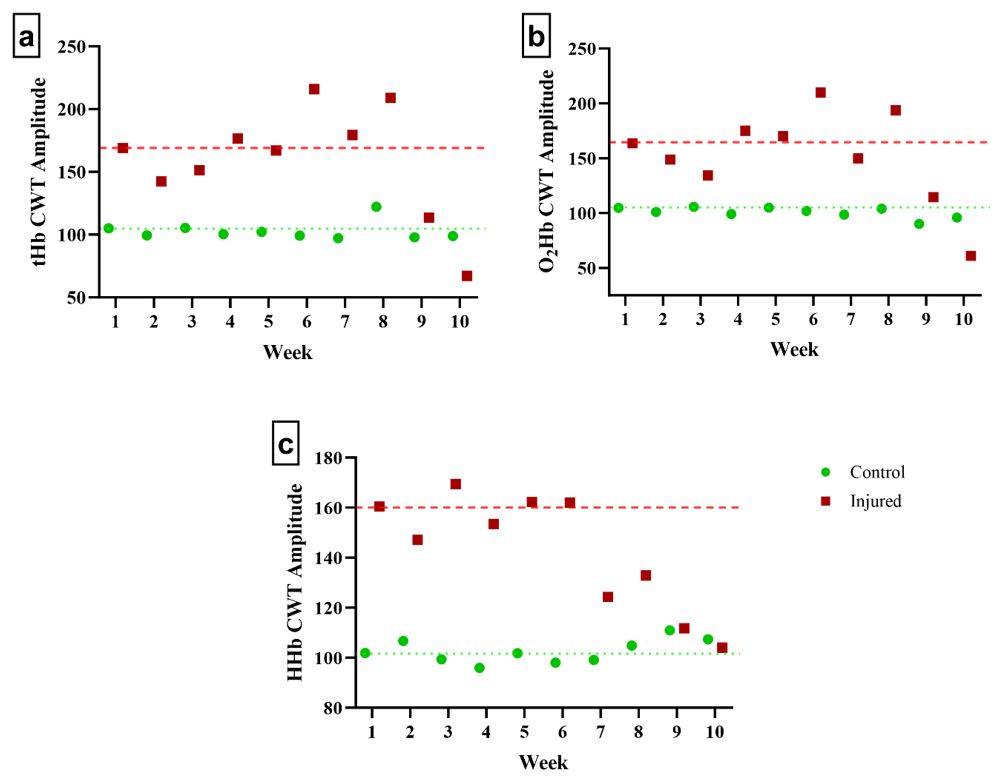
Figure 4: The continuous wavelet analysis (CWT) amplitudes averaged across all 4 optode in the 4x1 grid and averaged across the 60-s psychomotor vigilance test during each of the 10 weeks. The square reflects the Injured region of the temples cerebral cortex and the circle reflects the Control (uninjured opposing temple location of the cerebral cortex) for the fNIRS derived a) total hemoglobin responses (tHb); b) oxygenated hemoglobin response (O2Hb); and c) deoxygenated hemoglobin response (HHb). The dashed line represents the neuroimaging amplitude value measured within 72-hr from the Injured side of the brain while the dotted line represents the same timepoint for the Control region of the brain.
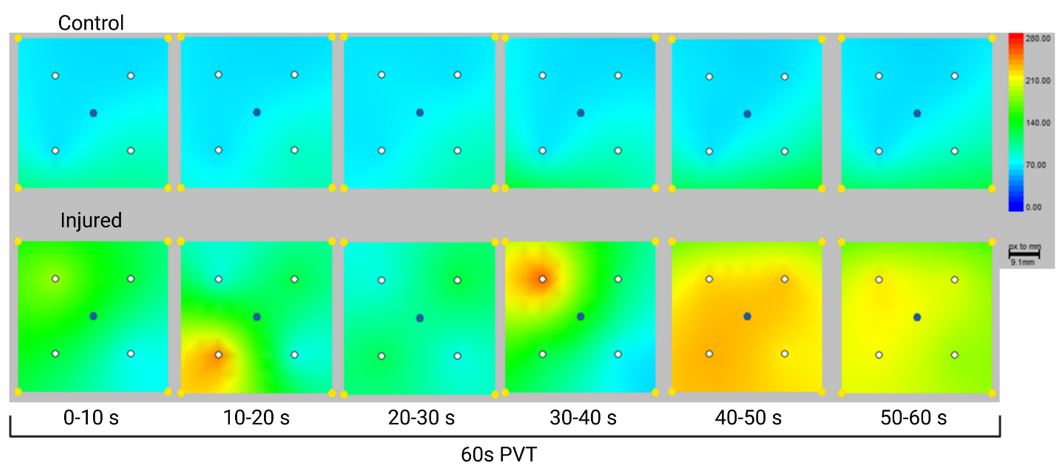
Figure 5: Heat map visual display of the 4x1 sensor grid wavelet for total hemoglobin hemodynamic response over the Control (unimpacted) and Injured (Impacted) regions of the patients head. The hand-eye coordination and motor control required to successfully perform the psychomotor vigilance test displays its ability to elicit greater detectable changes in the hemodynamic responses over the injured region.
functional Near-Infrared Spectroscopy: Neuroimaging: Neuroimaging the contralateral side of the brain as a Control was effective for identifying the hemodynamic and metabolic differences from the Injured region of the brain in our patient as a pre-injury image was unavailable. The relatively consistent tHbamp, HHbamp, and O2Hbamp metrics across the 10 weeks for the Control compared to the Injured side reflects a low neurophysiological load placed on the Control region of the brain typical of a non-mTBI patient during the 60-s PVT (Figure 4).2,26 Furthermore, Figure 5 illustrates the similarities in the neurophysiological load placed on the Control and Injured regions of the brain at the initial 0 to 10-s of the 60-s PVT, however, after 30-s of load a greater hemodynamic and metabolic responses occurred in the Injured but not the Control region of the brain. Thus, the 60-s PVT load placed on the brain was minimal enough to not impact the Control region of the brain while sufficient at stimulating a response from the Injured region of the brain. Therefore, the utilization of a Control region was effective and allowed us to develop a target recovery threshold for the Injured region of the brain to match the tHbamp, HHbamp, and O2Hbamp of the Control region.
The combined tHbamp, HHbamp, and O2Hbamp pattern of responses throughout the 10 weeks of recovery indicated maintenance of the patients Oxygen Extraction Fraction (OEF) in the Injured region of the brain. 32 The HHbamp and O2Hbamp ratio remained relatively constant in the Injured region of the brain with a concomitant increase in tHbamp which suggested that greater oxygenation utilization was required in the Injured region of the brain compared to the Control (Figure 4). The overall increased blood flow to the Injured region and increased metabolic demand, as indicated by the greater HHbamp, likely aimed to offset the mTBI associated Ca2+ overload within the patient. Increasing of the patients overall regional blood flow (tHbamp) to the Injured region may reflect a protective mechanism to avoid the catabolic effects of a Ca2+-induced intracellular dysregulation that has been shown to result in the overproduction of free radicals, activation of cell death signaling pathways and stimulation of inflammatory responses.33–35 That is, the fNIRS responses captured throughout the recovery of this patient tracked with the expected systemic physiological responses associated with the maintenance of OEF. Furthermore, it has been well established that mTBI’s result in mitochondrial dysregulation which result in a greater hemodynamic shift to the injured region of the brain to provide sufficient oxygen, dilution, and clearance rates for the metabolic byproducts.33,34 It is hypothesized that the combined mitochondrial dysregulation induced hemodynamic shift was likely the driving factor for the increased metabolic stress (HHbamp) while OEF further stimulated a greater hemodynamic flow to the Injured region of the brain to avoid further injury associated with a buildup of metabolic byproducts. The combined regional cerebral blood flow regulation pattern (tHbamp) and increased metabolic demand captured in this patient indicated that the damage to the patient’s brain was detectable using our fNIRS approach. Furthermore, the combined utilization of our CWT tHbamp, HHbamp, and O2Hbamp methodology allowed for identification of the hemodynamic shifts associated with the injury and increased load induced by the 60-s PVT.
In our patient, all fNIRS metrics that were elevated from weeks 1 to 8 improved to within Control values at week 9 and remained at the Control levels at week 10 (Figure 4). The tracked improvements in all fNIRS measures suggest that this patient’s recovery became physiologically improved at week 9, however, the patient reported minimal symptomology at rest at week 7 which cleared the patient to begin light physical activity. During weeks 7 and 8 the patient reported increased symptomology when exercising which was her rate limiter to perform physical activity. Considering the onset of the symptomology due to exercise and the fNIRS detected hemodynamic shifts associated with mitochondrial dysregulation coupled with OEF, the patient may have been less symptomatic if exercise was resumed at week 9.32,34 Specifically, the improved fNIRS and 60-s PVT metrics at week 9 were closely aligned with the Control and expected reaction time values, respectively, suggesting that our fNIRS approach could track the physiological recovery in this patient (Figure 4). Early physical activity in patients with mTBI’s has been linked to increase metabolic byproduct accumulation, greater hemodynamic shifts, potential reduced recovery rates, and increased symptomology.32,34,36,37 Thus, the elevated tHbamp, HHbamp, and OxyHbamp values at week 8 suggest that the physical activity performed at weeks 7 and 8 may have regressed recovery and induced additional dysfunction, lengthening recovery time. Therefore, further development of this non-invasive neuroimaging approach will provide clinicians with a useful assessment tool to make more informed decisions on the rate of recovery and activity a patient may be prescribed.
Limitations: This was an exploratory case study performed on a single patient to examine the clinical feasibility of the fNIRS and 60-s PVT analysis approach which will need greater refinement and development prior to clinical adoption. We acknowledge that greater data from a larger population should be studied to make this technology useable when real-time accounts or video footage of the injury site is unavailable. Thus, data from this study should not be applied to a broader patient pool until further studies focusing on the refinement and application of this approach is completed. The data from this case study does provide the foundational information needed to replicate the study methodology and highlights the relevance to the clinical community aiming to develop non-invasive mTBI monitoring devices.