The Applications and challenges of Probiotics/ Postbiotics in Cosmetic and personal care products
Shengquan Huang, Hailing Wang, Xiaolan Wang
Renhe Global (Shanghai) Big Health Research Institute Co., LTD. Guangfu West Road No. 2666, Putuo District, Shanghai 200333, P.R. China
*Corresponding author
*Shengquan Huang, Renhe Global (Shanghai) Big Health Research Institute Co., LTD. Guangfu West Road
No. 2666, Putuo District, Shanghai 200333, P.R. China
DOI: 10.55920/JCRMHS.2023.04.001151
Figure 1: The top 30 words used in the claims of probiotic products. Word cloud generated using a compilation of the shares of at least 50 products claiming to contain probiotics.
Numerous studies provide evidence of the benefits of specific postbiotics for skin health [11-13]. Meanwhile, anti-ageing mechanisms suggest strains can help regulate pH, reduce oxidative stress, protect from photoaging, and improve skin barrier function [14]. However, the cosmetic industry needs to be consistent and transparent in its labelling practices and direct efforts to generate more scientific evidence before making claims.
Topical delivery and formulation of probiotics
Not all applications for skincare for males or females require a local application. Orally administered probiotics have been demonstrated to affect the intestinal microbiome leading to a potential improvement in skin conditions such as atopic dermatitis, acne, and/or rosacea [15-18]. Early studies suggested that probiotic may be required during gestation and early life to reduce the incidence and adversity of atopic dermatitis [19], implying immune modulation and improving the maturing gut barrier function [20].
Freeze-drying of probiotic strains is common place. However, depending on the drying protectant used, final viability can vary. The most used protectants are skim milk, serum, trehalose, glycerol, betaine, adonitol, sucrose, glucose, lactose, and polyethylene glycol; these may not be compatible with the intended use of the product or the physicochemical characteristics of the formula [21]. When using this method, the strains should not be exposed to water; otherwise, they will prematurely rehydrate [22]. It is challenging for the cosmetic industry to create topical formulas that retain probiotic bacterial viability from production to the value chain and the consumer. Moisture would allow the dried organisms to hydrate and multiply or die, so oil-based formulations are needed. The question becomes how easily the organisms can emerge from the oil once placed on the skin and thence become metabolically active sufficient to deliver the probiotic effects required. In addition, many creams are not produced in sterile conditions; therefore, preservatives are often added with bactericidal and/or bacteriostatic effects. These potentially can affect the probiotic strain viability and inadvertently alter the recipient’s microbiota.
Regulation of probiotics is primarily concerned with safety. There is no specific requirement for commercializing probiotics, and products are regulated according to their final use, whether as a drug, medical device, food, dietary supplement, or cosmetic. The descriptiveness and level of documentation required to claim a cosmetic probiotic are substantially less than for one making drug claims in China and elsewhere. However, to maximize profit, some companies make disease or illness-alleviation statements associated with their cosmetic products, when this should be reserved for drugs or clinically proven supplements.
Due to safety concerns, cosmetic products are expected to have a low content of microorganisms. It is not a viable option for them to contain live bacteria, meaning that there cannot be a cosmetic that is a true probiotic. However, they can still have components sourced from probiotic strains that could be beneficial. These can constitute bacterial lysates, ferments, and filtrates, sometimes referred to as postbiotics, defined as a “preparation of inanimate microorganisms and/or their components that confers a health benefit on the target host. [23].” This definition does not include purified metabolites or components without cells, which should be listed following their chemical nomenclature. Filtrates without cell components are not considered postbiotics. However, depending on their composition, bacterial lysates and ferments might fit into this category.
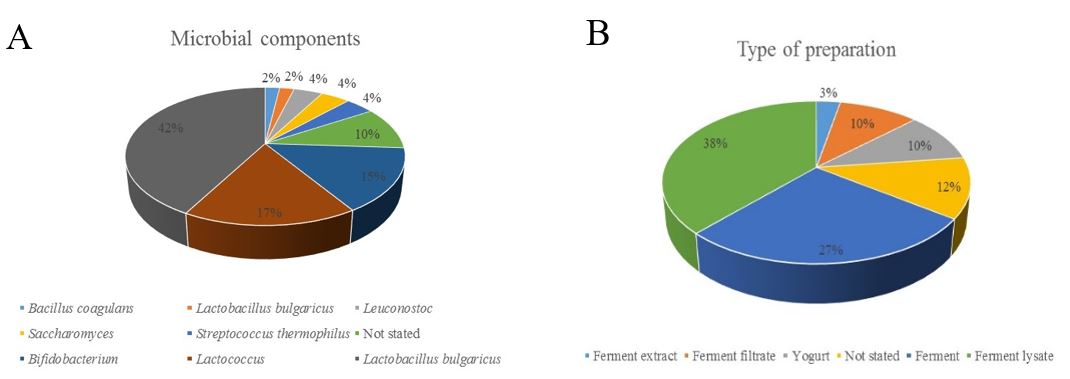
However, as shown in Figure 2, from the ingredients in the labels of at least the 50 cosmetic products we analyzed, none of them stated the strain designation of the microorganism included, and the only minority of the listed ingredients had the name of the species used. Those products listing probiotic use included it on their labels as “probiotic ferment lysate,” documented as an inactivated suspension of probiotics in an aqueous medium, which, as previously described, can improve barrier function as well as decrease skin sensitivity. Nonetheless, products containing this ingredient cannot be marketed as probiotics due to the absence of live bacteria.
With proper regulations in place, and potentially labelled as over-the-counter (OTC) drugs instead of cosmetics, there is value in using probiotics as topical treatments. Their antibacterial and immunomodulatory properties make them promising candidates to target skin ailments such as acne, psoriasis, and atopic dermatitis, and aid in wound healing [16, 18, 24-27]. Nonetheless, further human research and randomized clinical trials are still required to validate these potential uses.
Figure 2: Debrief of the ingredients listed in 50 cosmetic products. Panel A shows the frequency in which each microbial component was included. Panel B shows the frequency in which each type of preparation was used in the products analyzed.
Probiotics for Female Intimate Care
A abundance of lactobacilli mostly populates a healthy vaginal environment. Various triggers, from the use of douches and antibiotics to multiple sexual partners and influx of pathogens into the area, disrupt the homeostasis giving rise to bacterial vaginosis, urinary tract infections, candidiasis, and other conditions. This provided a rationale to supplement the urogenital tract with lactobacilli to restore a healthy state. Therefore, the vaginal administration of probiotic strains of Lactobacillus through suppositories or vaginal ovules has been explored [16]. A range of strains have been tested, including Limosilactobacillus (formerly Lactobacillus) Reuteri RC-14 and Lactobacillus crispatus CTV05 to reduce the recurrence of urinary tract infection (UTI) [28, 29], Lacticaseibacillus rhamnosus IMC 501 in combination with Lactobacillus paracasei IMC 502 to maintain vaginal homeostasis [30] and L. rhamnosus Lcr35 for B.V. and vulvovaginal candidiasis [31, 32].
Given the significant negative impact of antimicrobial therapy on the urogenital microbiota and failure to restore homeostasis, probiotic strains have been used in combination to help with recovery. These include Lactobacillus gasseri EN-153471 (EB01) for the management of B.V. [33] and L. rhamnosus GR-1 plus L. reuteri RC-14 in combination with antibiotics or antifungals [34]. Additional strains have become available in the American market with minimal clinical and scientific documentation [35]. The supplements are believed to function through an ascension from the rectal skin to the vagina, where they reduce pathogen ascension and inhibit and displace pathogens while also conferring antimicrobial defenses through the production of bioactive compounds such as lactic acid, hydrogen peroxide, and bacteriocins. Therefore, these are essentially cosmetic in action on the skin. Still, they are promoted through a higher level of regulation where some functional, structural, or even disease risk reduction claims can be made. The application of strains directly into the vagina using suppositories applied vaginally is approved in China. Other products are being delivered through coating tampons and pomades, but further evidence is required to confirm they are probiotics and benefit the host.
An emerging area is for probiotic strains to reduce urogenital malodor that significantly impacts the quality of life of women, especially in combination with bacterial vaginosis [36]. Many non-probiotic products such as vaginal douches, vinegar rinses, and fragrances claim to help reduce malodor. However, their efficacy is dubious, and they can increase the risk of infection, including sexual acquisition, by disrupting of the beneficial microbes [37, 38]. An advantage of an effective probiotic is its ability to grow and produce metabolites that degrade or neutralize malodorous compounds [39, 40]. Depending on the nature of such a product, it may have to be registered as an OTC drug and not as a cosmetic or personal care product. The development of topical gel containing probiotic lactobacilli is already underway, with promising results for treating vulvo-vaginal candidiasis [41].
It could be argued that applications in the urogenital tract do not strictly fall within the definition of a cosmetic intended to restore or improve a person’s appearance. Likewise, applications for reducing halitosis [42-44] may also not fit, depending on how‘appearance’ is defined and interpreted. Reducing acne symptoms with lactobacilli in a topical cream would work as a cosmetic [45].
- Siciliano RA, Reale A, Mazzeo MF, Morandi S, Silvetti T, Brasca M. Paraprobiotics: A New Perspective for Functional Foods and Nutraceuticals. Nutrients. 2021; 13: 1225.
- Nakatsuji, T.; Chiang, H.-I.; Jiang, S.B.; Nagarajan, H.; Zengler, K.; Gallo, R.L. The microbiome extends to subepidermal compartments of normal skin. Nat. Commun. 2013, 4, 1431.
- Matts, P.J. New insights into skin appearance and measurement. J. Invest. Dermatol. Symp. Proc. 2008, 13, 6–9.
- Guéniche, A.; Bastien, P.; Ovigne, J.M.; Kermici, M.; Courchay, G.; Chevalier, V.; Breton, L.; Castiel-Higounenc, I. Bifidobacterium longum lysate, a new ingredient for reactive skin. Exp. Dermatol. 2010, 19, 1–8.
- Klein, G.; Schanstra, J.P.; Hoffmann, J.; Mischak, H.; Siwy, J.; Zimmermann, K. Proteomics as a quality control tool of pharmaceutical probiotic bacterial lysate products. PLoS ONE 2013, 8, e66682
- Villa, E.; Garelli, V.; Braido, F.; Melioli, G.; Canonica, G.W. May we strengthen the human natural defenses with bacterial lysates? World Allergy Organ. J. 2010, 3, S17–S23.
- Mokrozub, V.V.; Lazarenko, L.M.; Sichel, L.M.; Babenko, L.P.; Lytvyn, P.M.; Demchenko, O.M.; Melnichenko, Y.O.; Boyko, N.V.; Biavati, B.; Digioia, D.; et al. The role of beneficial bacteria wall elasticity in regulating innate immune response. EPMA J. 2015, 6, 13.
- Mohammedsaeed, W.; Cruickshank, S.; McBain, A.J.; O’Neill, C.A. Lactobacillus rhamnosus GG lysate increases re-epithelialization of keratinocyte scratch assays by promoting migration. Sci. Rep. 2015, 5, 16147.
- Ankrah, N.Y.D.; May, A.L.; Middleton, J.L.; Jones, D.R.; Hadden, M.K.; Gooding, J.R.; LeCleir, G.R.;Wilhelm, S.W.; Campagna, S.R.; Buchan, A. Phage infection of an environmentally relevant marine bacterium alters host metabolism and lysate composition. ISME J. 2014, 8, 1089–1100.
- Genova, E.; Pelin, M.; Decorti, G.; Stocco, G.; Sergo, V.; Ventura, A.; Bonifacio, A. SERS of cells: What can we learn from cell lysates? Anal. Chim. Acta 2018, 1005, 93–100.
- Navarro-López, V.; Martínez-Andrés, A.; Ramírez-Boscá, A.; Ruzafa-Costas, B.; Núñez-Delegido, E.; Carrión-Gutiérrez, M.A.; Prieto-Merino, D.; Codoñer-Cortés, F.; Ramón-Vidal, D.; Genovés-Martínez, S.; et al. Efficacy and safety of oral administration of a mixture of probiotic strains in patients with psoriasis: A randomized controlled clinical trial. Acta Derm. Venereol. 2019, 99, 1078–1084.
- Yu, Y.; Dunaway, S.; Champer, J.; Kim, J.; Alikhan, A. Changing our microbiome: Probiotics in dermatology. Br. J. Dermatol. 2020, 182, 39–46.
- Korpela, K.; Salonen, A.; Vepsäläinen, O.; Suomalainen, M.; Kolmeder, C.; Varjosalo, M.; Miettinen, S.; Kukkonen, K.; Savilahti, E.; Kuitunen, M.; et al. Probiotic supplementation restores normal microbiota composition and function in antibiotic-treated and in caesarian-born infants. Microbiome 2018, 6, 182.
- Sharma, D.; Kober, M.M.; Bowe,W.P. Anti-aging effects of probiotics. J. Drugs Dermatol. 2016, 15, 9–12.
- Jung, G.W.; Tse, J.E.; Guiha, I.; Rao, J. Prospective, randomized, open-label trial comparing the safety, efficacy, and tolerability of an acne treatment regimen with and without a probiotic supplement and minocycline in subjects with mild to moderate acne. J. Cutan Med. Surg. 2013, 17, 114–122.
- Knackstedt, R.; Knackstedt, T.; Gatherwright, J. The role of topical probiotics in skin conditions: A systematic review of animal and human studies and implications for future therapies. Exp. Dermatol. 2020, 29, 15–21.
- Nichols RG, Peters JM, Patterson AD. Interplay between the host, the human microbiome, and drug metabolism. Hum Genomics. 2019; 13(1):27.
- Siddiqui R, Makhlouf Z, Khan NA. The increasing importance of the gut microbiome in acne vulgaris. Folia Microbiol (Praha). 2022. doi: 10.1007/s12223-022-00982-5.
- Kalliomäki, M.; Salminen, S.; Poussa, T.; Arvilommi, H.; Isolauri, E. Probiotics and prevention of atopic disease: 4-year follow-up of a randomized placebo-controlled trial. Lancet 2003, 361, 1869–1871.
- Isolauri, E.; Kalliomaki, M.; Laitinen, K.; Salminen, S. Modulation of the maturing gut barrier and microbiota: A novel target in allergic disease. Curr. Pharma. Des. 2008, 14, 1368–1375.
- Hubálek, Z. Protectants used in the cryopreservation of microorganisms. Cryobiology 2003, 46, 205–229.
- Morgan, C.A.; Herman, N.; White, P.A.; Vesey, G. Preservation of microorganisms by drying; a review. J. Microbiol. Methods 2006, 66, 183–193.
- Marco, M.L.; Sanders, M.E.; Gänzle, M.; Arrieta, M.C.; Cotter, P.D.; De Vuyst, L.; Hill, C.; Holzapfel, W.; Lebeer, S.; Merenstein, D.; et al. The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on fermented foods. Nat. Rev. Gasteroenterol. Hepatol. 2021.
- Jung, G.W.; Tse, J.E.; Guiha, I.; Rao, J. Prospective, randomized, open-label trial comparing the safety, efficacy, and tolerability of an acne treatment regimen with and without a probiotic supplement and minocycline in subjects with mild to moderate acne. J. Cutan Med. Surg. 2013, 17, 114–122.
- Nichols RG, Peters JM, Patterson AD. Interplay between the host, the human microbiome, and drug metabolism. Hum Genomics. 2019; 13(1):27.
- Rather, I.A.; Bajpai, V.K.; Huh, Y.S.; Han, Y.-K.; Bhat, E.A.; Lim, J.; Paek,W.K.; Park, Y.-H. Probiotic Lactobacillus sakei proBio-65 extract ameliorates the severity of imiquimod induced psoriasis-like skin inflammation in a mouse model. Front. Microbiol. 2018, 9, 1021.
- Sonal Sekhar, M.; Unnikrishnan, M.K.; Vijayanarayana, K.; Rodrigues, G.S.; Mukhopadhyay, C. Topical application/formulation of probiotics: Will it be a novel treatment approach for diabetic foot ulcer? Med. Hypotheses. 2014, 82, 86–88.
- Reid, G.; Bruce, A.W.; Fraser, N.; Heinemann, C.; Owen, J.; Henning, B. Oral probiotics can resolve urogenital infections. FEMS Immunol. Med. Microbiol. 2001, 30, 49–52.
- Stapleton, A.E.; Au-Yeung, M.; Hooton, T.M.; Fredricks, D.N.; Roberts, P.L.; Czaja, C.A.; Yarova-Yarovaya, Y.; Fiedler, T.; Cox, M.; Stamm, W.E. Randomized, placebo-controlled phase 2 trial of a Lactobacillus crispatus probiotic given intravaginally for prevention of recurrent urinary tract infection. Clin. Infect. Dis. 2011, 52, 1212–1217.
- Verdenelli, M.C.; Cecchini, C.; Coman, M.M.; Silvi, S.; Orpianesi, C.; Coata, G.; Cresci, A.; Di Renzo, G.C. Impact of probiotic SYNBIO® administered by vaginal suppositories in promoting vaginal health of apparently healthy women. Curr. Microbiol. 2016, 73, 483–490.
- Rossi, A.; Rossi, T.; Bertini, M.; Caccia, G. The use of Lactobacillus rhamnosus in the therapy of bacterial vaginosis. Evaluation of clinical efficacy in a population of 40 women treated for 24 months. Arch. Gynecol. Obstet. 2010, 281, 1065–1069.
- Petricevic, L.;Witt, A. The role of Lactobacillus casei rhamnosus Lcr35 in restoring the normal vaginal flora after antibiotic treatment of bacterial vaginosis. Gen. Gynaecol. 2008, 1369–1374.
- Larsson, P.-G.; Brandsborg, E.; Forsum, U.; Pendharkar, S.; Andersen, K.K.; Nasic, S.; Hammarström, L.; Marcotte, H. Extended antimicrobial treatment of bacterial vaginosis combined with human lactobacilli to find the best treatment and minimize the risk of relapses. BMC Infect. Dis. 2011, 11, 223.
- Anukam, K.; Osazuwa, E.; Ahonkhai, I.; Ngwu, M.; Osemene, G.; Bruce, A.W.; Reid, G. Augmentation of antimicrobial metronidazole therapy of bacterial vaginosis with oral probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14: Randomized, double-blind, placebo-controlled trial. Microbes Infect. 2006, 8, 1450–1454.
- Puebla-Barragan S, Reid G. Probiotics in Cosmetic and Personal Care Products: Trends and Challenges. Molecules. 2021; 26(5):1249.
- Bilardi, J.E.; Walker, S.; Temple-Smith, M.; McNair, R.; Mooney-Somers, J.; Bellhouse, C.; Fairley, C.K.; Chen, M.Y.; Bradshaw, C. The burden of bacterial vaginosis: Women’s experience of the physical, emotional, sexual and social impact of living with recurrent bacterial vaginosis. PLoS ONE 2013, 8, e74378.
- Fashemi, B.; Delaney, M.L.; Onderdonk, A.B.; Fichorova, R.N. Effects of feminine hygiene products on the vaginal mucosal biome. Microb. Ecol. Heal. Dis. 2013, 24.
- Brotman, R.M.; Klebanoff, M.A.; Nansel, T.R.; Andrews,W.W.; Schwebke, J.R.; Zhang, J.; Yu, K.F.; Zenilman, J.M.; Scharfstein, D.O. A longitudinal study of vaginal douching and bacterial vaginosis—a marginal structural modeling analysis. Am. J. Epidemiol. 2008, 168, 188–196.
- McMillan, A.; Rulisa, S.; Sumarah, M.; Macklaim, J.M.; Renaud, J.; Bisanz, J.E.; Gloor, G.B.; Reid, G. A multi-platform metabolomics approach identifies highly specific biomarkers of bacterial diversity in the vagina of pregnant and non-pregnant women. Sci. Rep. 2015, 5, 1–14.;
- Puebla-Barragan, S.; Renaud, J.; Sumarah, M.; Reid, G. Malodorous biogenic amines in Escherichia coli-caused urinary tract infections in women—a metabolomics approach. Sci. Rep. 2020, 10, 9703.
- Donders, G.; Bellen, G.; Oerlemans, E.; Claes, I.; Ruban, K.; Henkens, T.; Kiekens, F.; Lebeer, S. The use of 3 selected lactobacillary strains in vaginal probiotic gel for the treatment of acute Candida vaginitis: A proof-of-concept study. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1551–1558.
- Iwamoto, T.; Suzuki, N.; Tanabe, K.; Takeshita, T.; Hirofuji, T. Effects of probiotic Lactobacillus salivarius WB21 on halitosis and oral health: An open-label pilot trial. Oral Surgery, Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 201–208.
- Masdea, L.; Kulik, E.; Hauser-Gerspach, I.; Ramseier, A.; Filippi, A.; Waltimo, T.; Masdea, L.; Kulik, E.; Hauser-Gerspach, I.; Ramseier, A.; et al. Antimicrobial activity of Streptococcus salivarius K12 on bacteria involved in oral malodour. Arch. Oral Biol. 2012, 57, 1041–1047.
- Burton, J.; Chilcott, C.; Moore, C.; Speiser, G.; Tagg, J. A preliminary study of the effect of probiotic Streptococcus salivarius K12 on oral malodour parameters. J. Appl. Microbiol. 2006, 100, 754–764.
- Lebeer, S.; Oerlemans, E.; Claes, I.; Wuyts, S.; Henkens, T.; Spacova, I.; van den Broek, M.; Tuyaerts, I.;Wittouck, S.; De Boeck, I.; et al. Topical cream with live lactobacilli modulates the skin microbiome and reduce acne symptoms. bioRxiv. 2018.