Primary Breast Angiosarcoma: A case report and literature review
Stathaki Martha1, Metaxas Georgios1, Christodoulidou Ioulia1, Gogkou Haralambos1, Skarpidi Evangelia2, Mantzouki Christina1, Dimou Evelina1 and Armakolas Athanasios3*
1Third Surgical Clinic, Elena Venizelos Hospital, Athens, Greece
2MICROMEDICALABS Diagnostic Laboratory, Athens, Greece
3Physiology Laboratory, Medical School, National and Kapodestrian University of Athens, Greece
*Corresponding author
*Athanasios Armakolas, Physiology Laboratory, Medical School, National and Kapodestrian University
of Athens, Greece
DOI: 10.55920/JCRMHS.2023.03.001137
Figure 1: Mammogram, ultrasound and breast MRI: A. Spot compression mammogram with non-specific findings. B. Determination of a diffuse area of increased echo texture by ultrasonographic imaging. C. MRI indicates the presence of a large lesion of the superior inner quadrant of the left breast exhibiting persisting and plateau enhancement kinetics.
A second look US scan with multiple guided core needle biopsies was performed (fig. 1c). Pathology described a highly vascular tissue sample, vascular endothelial cells with mild cellular atypia, presence of hob nail like cells, and a Ki-67 of 20-30%. Immunostaining assay for vascular endothelial marker CD31 and markers FLI1 and ERG was markedly positive. A diagnosis of a low to intermediate grade angiosarcoma was concluded. Due to the discordance of the radiology and pathology reports, the sample was referred to a second pathologist who confirmed the diagnosis of well differentiated PBAS. Staging with abdominal, chest and head CT scans did not reveal distant metastases (fig. 2a and b).
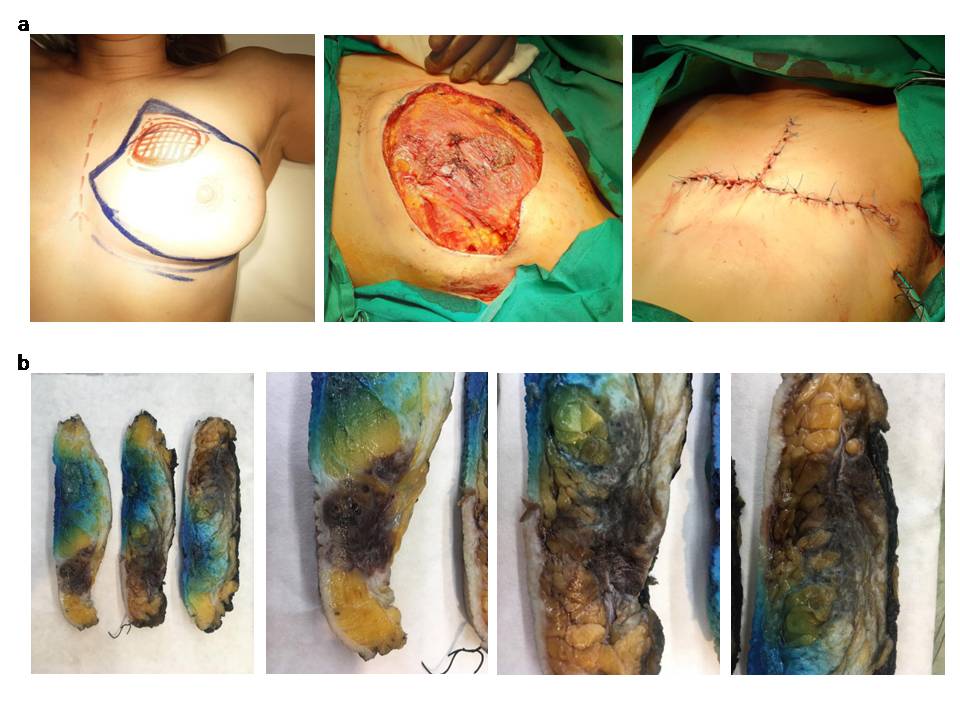
Figure 2: Surgical procedure: A. Surgical planning and mastectomy with partial excision of the pectoralis major muscle. B. Cross section of the resected breast showing a poorly circumscribed tumour that extends to the overlying skin.
Following a detailed discussion, the patient was submitted to a right mastectomy and sentinel lymph node biopsy. A superficial layer of pectoralis major fibers underlying the area of concern was removed en block with the breast. Gross examination of the specimen confirmed the presence of a 5,8cm tumor with irregular margins within the breast parenchyma. On cut section the mass had a dark brown to pink color and soft consistency. Microscopically, the tumor was composed of an intricate anastomotic network of vascular channels, with papillary formations, mild to moderate cellular atypia and a mitotic index of 8/mm². Necrosis was absent. The closest radial margin was > 15mm, while the posterior margin though close was negative and there was no infiltration towards the muscle fibers. The 2 sentinel lymph nodes were also negative. The final diagnosis was low/intermediate grade breast angiosarcoma (T3 N0M0) (fig. 3a, b).
Figure 3: Immunohistochemical analysis of the tunour: A. H& E staining. The tumour consisted of anastomosing vascular spaces, lined by cells with mild to moderate atypia. In the absence of high grade features elsewhere, this growth patern may mimic a benign vascular lesion. B. Neoplastic vascular spaces infiltrating a breast lobule.
The oncology board suggested adjuvant radiotherapy only which the patient received in 30 cycles. At 15 months postoperatively the patient remains disease free.
- Abdou Y., Elkhanany A., Attwood K., et al. Primary and secondary breast angiosarcom: single center report and meta-analysis. Breast C. Res. & Treat. (2019) 178:523-533.
- Nascimento AF., Raut CP., Fletcher CD. Primary angiosarcoms of the breast: clinicopathologic analysis of 49 cases, suggesting that grade is not prognostic. Am. J. Surg. Pathol. (2008) 32:1896-1904.
- Wei Y., Yang Xi, Gao L., et al. Differencies in potential key genes and pathways between primary and radiation-associated angiosarcoma of the breast. Transitional Oncology (2022) 101385.
- Esposito E., Avino F., Raimondo dG., et al. Angiosarcoma of the breast, the unknown- a review of the current leterature. Transl. Cancer Res. (2019); 8 (Suppl): 510-517.
- Wang XY., Jakowski J., Tawfik Ow., et a.l. Angiosarcoma of the breast: a clinicopathologic analysis of cases from the last 10 years. |Ann. Ciagn. Pathol. (2009); 13: 147-150.
- Rohan VS., Hanji AM., Patell JJ., et al. Priamary angiosarcoma of the breast in a postmenopausal patient. J. Cancer Res. Ther. (2010); 6: 120-122.
- Beenu V., Pooja D., Santosh D., et al. Primary angiosarcoma of the breast : a case report. Rad. Case (2019) Feb; 13(2):15-25.
- Dhirenda NS., Gopalakrishnan G., Debasis N., et al. Primary mammary angiosarcoma: Literature review. Cureus (2020); 12(6): e8589.
- Mahdi Y., Rouas L., Malihy A., et al. Diagnostic difficulties of primary angiosarcoma of the breast: a case report. Journal of Med. Case Rep. (2018) 12¨228-233.
- Dashensky BZ., Charnoff-Katz K., Shin SJ., et al. A case of primary breast angiosarcoma. Radiol. |Case Rep. (2015) Nov 6; 8(3):741.
- Yang WT., Hennessy BT., Dryden MJ., et al. Mammary angiosarcomas: imaging findings in 24 patients. Radiology (2007); 242:725-734.
- Arora TK., Terracina KP., Soong J., et al. Primary and secondary angiosarcoma of the breast. Gland Surg. (2014) Feb;3(1): 28-34.
- O`Neill AC., D`Acry C., McDermott E., et al. Magnetic resonance imaging appearances in primary and secondary angiosarcoma of the breast. J. Med. Imaging Radiat. Oncol. (2014); 58:208-212.
- Im S., Chae BJ., Kim SH., et al. Primary angiosarcoma of the breast: a case report. Int. J. Clin. Exp. Pathlol. (2019); 12(2): 664-668.
- Chen K.T.K., Kerkegaard D.D., Bocian J.J.: Angiosarcoma of the breast. Cancer (1980);46: 268-271.
- Lim R.F., GoeiR..Best cases from the AFIP: angiosarcoma of the breast. Radiographics (2007); Oct; 27(1): 125-130.
- Haroon S., Faridi N., Lodhi F.R.. Primary angiosarcoma of breast. J. coll. Physicians Surg. Pak. (2013); May;23(5): 356-358.
- Desbiens C., Hogue J.C., levesqueY..Primary breast angiosarcoma: Avoiding a common trap. Case Rep. Oncol. Med. (2011);2011:517047.
- Bennani A., Chbani L., LamchahabM..Primary angiosarcoma of the breast: a case report. Diagn. Pahtol. (2013) Apr 22;8:66.
- Pandley M., Martin M.G.. Primary angiosarcoma of the breast: a case report and review of literature. World J. Oncol.(2014) Jun;5(3):144-148.
- Glazebrook K.N., Magut M.J., Reynolds C.. Angiosarcoma of the breast. Am. J. Roentgenol. (2008) Feb;190(2): 533-538.
- Jesinger R.A., Lattin G.E., Ballard E.A.. et al. Vascular abnormalities of the breast: arterial and venous disorders, vascular masses, and mimic lesions with radiologic- pathologic correlation. Radiographics (2011) Nov-Dec;31(7): 117-136.
- Ohta M., Tokuda Y., Kuge S., et al. A case of angiosarcoma of the breast. Jpn. J. Clin., Oncol. (1977) Apr;27(2): 91-94.
- Marchant L.K., Orel S.G., Perez-Jaffe L.A. et al.. Bilateral angiosarcoma of the breast on MR imaging. AM. J. Roentgenol. (1997) Oct;169(4): 1009-1010.
- Dashevsky B.Z., Charnoff-Katz K., Shin S.J. et al.. A case of primary breast angiosarcoma. Radiol. Case Rep. (2015); Nov 6;8(3): 741.
- Lim S.Z., Ong K.W., Tan B.K., et al.. Sarcoma of the breast: an update on a rare entity. J. Clin. Pathol. (2016);69: 373-381.
- Arora T., Terracina K., Soong J., et al..Primary and secondary angiosarcma of the breast. Gland Surg. (2014);3(1): 28-34.
- Shiraki E., Yookija K., Takahiro S. and Tsuyuki S. Two cases of breast angiosarcoma after breast conserving surgery. Surg. Case Reports(2020);6:81.
- Fugisawa Y., FujimuraT, Matsushita S. et al. The efficacy of eribulin mesylate for patients with cutaneouw Http angiosarcoma previously treated with taxane: a multi-center, prospective observatinal study. Br. J. Dermatol. (2020) https//doi.org/10.1111/bjd.19042.
- Johnstone P.A., Pierce L.J., Merino M.J., et al.. Primary soft tissue sarcomas of the breast: local-regional control with post-operative radiotherapy. Int. J. Radiat. Oncol. Bio. Phys. (1993);27: 359-361.
- Hodgson N.C., Bowen-Wells C., Moffat F., et al.. Angiosarcomas of the breast: A review of 70 cases. Am. J. Clin. Oncol. (2007);30: 570-573.
- Kim Y.J., Ryu J.M., Lee S.K., et al.. Primary angiosarcoma of the breast: A single-center retrospective study in Korea. Curr. Oncol. (2022);29: 3272-3281.
- Kunkiel M., Maczkiewicz M., Jagiello-Gruszfeld A., et al.. Primary angiosarcoma of the breast-series of 11 consecutive case-as single center experience. Curr. Oncol. (2018);25:e50e53.
- Abdou Y., Elkhanany A., Attwood K., et al.. Primary and secondary breast angiosarcoma: Single center report and meta-analysis. Brest Cancer Res. Treat. (2019);178: 523-533.
- Johnson C.M., GarguiloG.A..andiosarcoma of the breast: A case report and literature review. Curr. Surg. (2002);59: 490-494.
- Arora TK., Terracina KP., Soong J., et al. Primary and secondary angiosarcoma of the breast. Gland Surg. (2014) Feb;3(1): 28-34.
- Masai K., Konoshita T., Jimbo K., et al. Clinicopathological features of breast angiosarcoma. Breast Cancer(2016);23(5):718-723.
- Sasahara A., Masahiko T., Hayasi K., et al..A case or primary breast angiosarcoma with multiple discontinuous small lesions. Surg. Case Rep.(2019);5:157.
- Bousquet G., Confavreux C., Magne N., et al. Outcome and prognostic factors in breast sarcoma, a multicenter study from the rare cancer network. Radiother. Oncol.92007);85(3):355-361.
- Pasta V., Monti M., Cialini M., et al. Primitive sarcoma of the breast: new insight on the proper surgical management. J. Exp. Clin. Cancer Res. (2015);34:72.
- Adem C., Reynolds C., Ingle J.N., et al. Primary breast sarcoma: clinicopathologic series from Mayo Clinic and review of the literature. Br. J. Cancer (2004);91(2):237-241.
- Losnoff J.E., Jaber S., Esuba M., et al. Primary angiosarcoma of the breast: do enlarged axillary nodes matter? Breast J.,(2006);12:371-374.
- Kunkiel M., Maczkiewidz M., Jagiello-Gruszfeld A., et al. Primary angiosarcoma of the breast-series of 11 consecutive dasew.- a single center experience. Curr. Oncol.(2018);25(1):E50-53.
- Andrade de Mello R., Figueiredo P., Marques M., et al. Concurrent breast stroma sarcoma and breast carcinoma: a case report. J. Med. Case Rep.(2010);4:414.