Celiac patient with new episodes of Diahrrea:What could it be?
Joana Dias Antunes1, Ivo Barreiro1, Luísa Loureiro2, Abílio Gonçalves2
1Internal Medicine Resident at Department of Internal Medicina, Hospital Distrital da Figueira da Foz
2Medical specialist in Internal Medicine at Department of Internal Medicina, Hospital Distrital da Figueira da Foz
*Corresponding author
*Joana Antunes, MD, Internal Medicine Resident at Department of Internal Medicina, Hospital Distrital
da Figueira da Foz, Portugal
DOI: 10.55920/JCRMHS.2022.02.001072
Figure 1: Subtotal villous atrophy with crypt hyperplasia and intra-epithelial lymphocytosis.
Figure 2: Colonic mucosa shows infiltration of the epithelium by multiple lymphocytes.
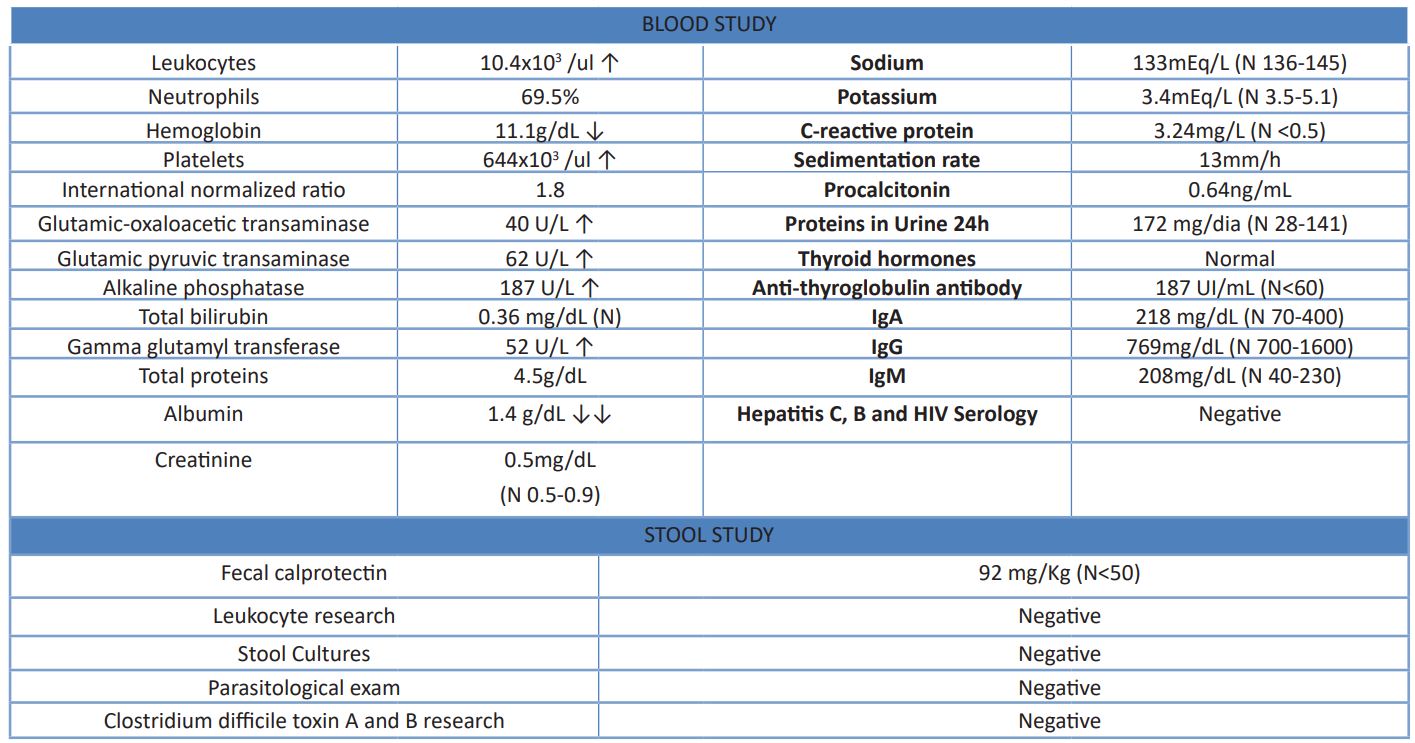
Table 1: Analytical study carried out on admission
- Arasaradnam RP, Brown S, Forbes A, Fox MR, Hungin P, Kelman L et al. Guidelines for the investigation of chronic diarrhoea in adults: British Society of Gastroenterology, 3rd edition. Gut. 2018; 67:1380–1399.
- Villanueva MS, Alimi Y. Microscopic colitis (lymphocytic and collagenous), eosinophilic colitis, and celiac disease. Clin Colon Rectal Surg. 2015; 28(2):118-126.
- Lauret E, Rodrigo L. Celiac disease and autoimmune-associated conditions. Biomed Res Int. 2013; 2013:127589.
- Barta Z, Zold E, Nagy A, Zeher M, Csipo I. Celiac disease and microscopic colitis: a report of 4 cases. World J Gastroenterol. 2011; 17(16):2150-2154.
- Stewart M, Andrews CN, Urbanski S, Beck PL, Storr M. The association of coeliac disease and microscopic colitis: a large population-based study. Aliment Pharmacol Ther. 2011; 33(12):1340-9.