Gold (III)-Gadolinium (II) Complex – Hybrid Nanoparticles: From Synthesis to Nanomedicine and Imaging Applications
Fatima Aouidat1, Memona Khan1, Xiaowu Li3-4*, Frederik Tielens5, Jolanda Spadavecchia1-4*
1CNRS, UMR 7244, CSPBAT, Laboratoire de Chimie, Structures et Propriétés de Biomatériaux et d’Agents Thérapeutiques Université Paris 13, Sorbonne Paris Cité, Bobigny, France.
3Department of hepato-biliary surgery, Shenzhen University General Hospital,Shenzhen, China
4Department of hepato-biliary surgery, Shenzhen University Clinical Medical Academy, Shenzhen, China
5General Chemistry (ALGC), Vrije Universiteit Brussel (Free University BrusselsVUB), Pleinlaan 2, 1050 Brussel, Belgium.
*Corresponding author
*Jolanda Spadavecchia, 1CNRS, UMR 7244, CSPBAT, Laboratoire de Chimie, Structures et Propriétés de Biomatériaux et d’Agents Thérapeutiques Université Paris 13, Sorbonne Paris Cité, Bobigny, France
DOI: 10.55920/JCRMHS.2022.02.001069
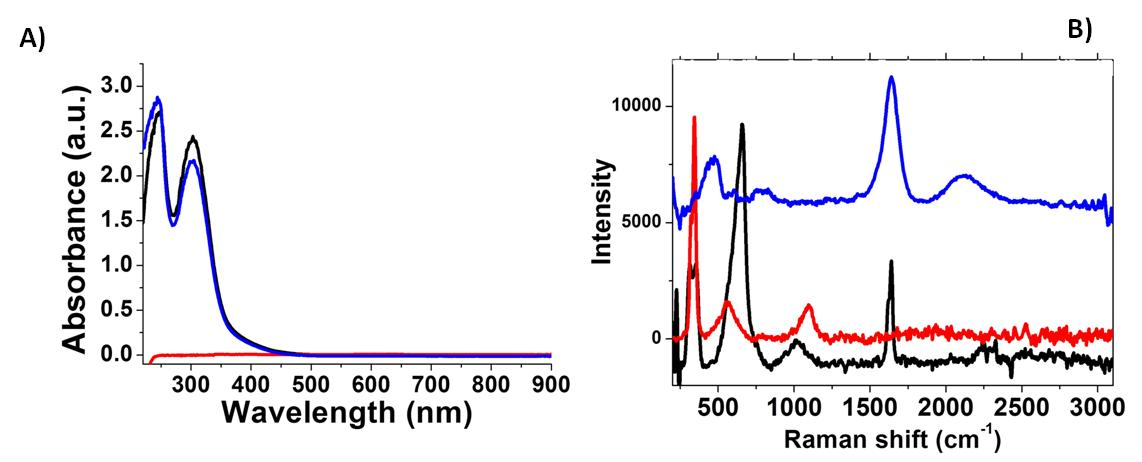
Figure 1 : P(A) UV-Vis absorption of HAuCl4*3H2O (black line), GdCl3*6H2O (red line); Au-Gd complex (blue line) in the range 200-900 nm (B) Raman spectra of Gd-Au complex (blue line) in the range 200-3000 cm-1, compared to HAuCl4*6H2O (black line), GdCl3*6H2O (red line) as controls. Experimental conditions: λexc = 785 nm; laser power 20 mW; accumulation time 180s./em>
Scheme 1: Schematic of proposed mechanism of GdCl3-AuCl4-reduction by complexation and particle formation (Gd(II) AuNPs) in the presence of polymer and sugar as surfactants (Please note that drawings are not in scale and are not intended to be representative of the full samples composition and stoichiometry).
Scheme 1: Schematic of proposed mechanism of GdCl3-AuCl4-reduction by complexation and particle formation (Gd(II) AuNPs) in the presence of polymer and sugar as surfactants (Please note that drawings are not in scale and are not intended to be representative of the full samples composition and stoichiometry).
Figure 2 :(A) UV-Vis absorption of Gd- Au complex (purple line) and after staking with PEG diacide (red line), COL (blue line) and CHIT (green line) and NP3 (black line) in the range 200-900nm. (A1) zoom oft he range 200-400 nm ; (A2) zoom oft he range 200-400 nm.
Figure 3: (A) TEM images of Gd (II) AuNPs (B) normalized UV-Vis absorption of NP1 (red line), NP2 ( blue line) and NP3 ( green line ) in the range 300-900 nm and (C) Raman spectra products compared to free GdCl3*6H20 as control. (A) Scale bars: 200 nm; 50 nm ; 20 nm. (D) Raman spectra. Experimental conditions: λexc = 785 nm; laser power 20 mW; accumulation time 180s.
Figure 4: The square planar (AuX4)+ precursor and the GdX3 precursor with X = Cl or OH.
Figure 5: (graph right) Interaction energy DEint as a function of the Cl substitution around Au and Gd for the complex HAu(OH)4-xClx and Gd(OH)3-yCly. Energies in eV. The values on the x axes correspond to the x values while the numbers in the legend correspond to the y values. The DEint for y = 1 are connected with a red line. a) Geometry of the most stable HAu (OH)4-xClx···Gd(OH)3-yCly complex. b)Geometry of the least stable HAu(OH)4-xClx···Gd(OH)3-yCly complex, showing a distorted trigonal geometry for Gd and a hydrogen bond between the Au and the Gd complex. The distance between both metal centers is 2.35 Å – 3.06 Å for structure a and b, respectively.
Figure 6: A) Viability by Alamar Blue assay of TIB-75 (hepatocytes) cell lines after incubation with Gd -Au nanoparticles (NP1-NP2-NP3); B) Examples of MRI images of T1 and T2 weighting recorded at 7T and the corresponding r1 and r2(respectively 20.0 and 8,8 mM-1.s-1), r1/r2 = 2.3) relaxivities values showing the efficient T1 type features of the contrast agent for MRI at 7T.
Table 1:Values of relaxivities r1 and r2 are expressed in (mM-1.s-1) measured at 7T, and corrected (second lines) from ICP AES elementary analysis (IPG ICP AES Facility, Paris); r2/r1 ratio are calculated to evidence the T1 type MRI contrast agent (>1).
Figure 8: Example of Dynamic Contrast Enhanced (DCE) MRI biodistribution in vivo before and after injection of v=100ul of solution of Gd -Au nanoparticules (NP3) during 48h post injection. Enhancement of the signal visualized the uptake of the NP in the organ and the disappearance of the signal showed the clearance.
Table 2: Timings of biodistribution of the NP3 measured in vivo from DCE MRI in mice.
[1] aG. G. Genchi, A. Marino, C. Tapeinos, G. Ciofani, Frontiers in Bioengineering and Biotechnology 2017, 5, 80; bS. Marchesan, M. Prato, ACS Medicinal Chemistry Letters 2013, 4, 147-149.
[2] aO. N. Oliveira, R. M. Iost, J. R. Siqueira, F. N. Crespilho, L. Caseli, ACS Applied Materials & Interfaces 2014, 6, 14745-14766; bO. N. Oliveira, Jr., R. M. Iost, J. R. Siqueira, Jr., F. N. Crespilho, L. Caseli, ACS Appl Mater Interfaces 2014, 6, 14745-14766.
[3] aS. K. Nune, P. Gunda, P. K. Thallapally, Y.-Y. Lin, M. L. Forrest, C. J. Berkland, Expert opinion on drug delivery 2009, 6, 1175-1194; bR. D. Mehlenbacher, R. Kolbl, A. Lay, J. A. Dionne, Nature Reviews Materials 2017, 3, 17080.
[4] K. C. Leung, S. Xuan, Chemical record (New York, N.Y.) 2016, 16, 458-472.
[5] J. F. Longevial, K. El Cheikh, D. Aggad, A. Lebrun, A. van der Lee, F. Tielens, S. Clement, A. Morere, M. Garcia, M. Gary-Bobo, S. Richeter, Chemistry 2017, 23, 14017-14026.
[6] H. S. Choi, J. V. Frangioni, Molecular imaging 2010, 9, 291-310.
[7] T. Yasui, N. Kaji, Y. Baba, Annual review of analytical chemistry (Palo Alto, Calif.) 2013, 6, 83-96.
[8] M. W. Bourassa, L. M. Miller, Metallomics : integrated biometal science 2012, 4, 721-738.
[9] S. S. Teske, C. S. Detweiler, International Journal of Environmental Research and Public Health 2015, 12, 1112-1134.
[10] A. M. Smith, S. Nie, Accounts of chemical research 2010, 43, 190-200.
[11] aC.-T. Yang, K.-H. Chuang, MedChemComm 2012, 3, 552-565; bE. Garanger, S. A. Hilderbrand, J. T. Blois, D. E. Sosnovik, R. Weissleder, L. Josephson, Chemical communications (Cambridge, England) 2009, 4444-4446.
[12] H. Wang, S. Zhang, X. Tian, C. Liu, L. Zhang, W. Hu, Y. Shao, L. Li, Scientific Reports 2016, 6, 34367.
[13] M. Rodriguez-Castillo, G. Lugo-Preciado, D. Laurencin, F. Tielens, A. van der Lee, S. Clement, Y. Guari, J. M. Lopez-de-Luzuriaga, M. Monge, F. Remacle, S. Richeter, Chemistry 2016, 22, 10446-10458.
[14] M. Rodríguez-Castillo, D. Laurencin, F. Tielens, A. van der Lee, S. Clément, Y. Guari, S. Richeter, Dalton Transactions 2014, 43, 5978-5982.
[15] aR. Sethi, J. S. Ananta, C. Karmonik, M. Zhong, S. H. Fung, X. Liu, K. Li, M. Ferrari, L. J. Wilson, P. Decuzzi, Contrast media & molecular imaging 2012, 7, 10.1002/cmmi.1480; bH. Wang, S. Zhang, X. Tian, C. Liu, L. Zhang, W. Hu, Y. Shao, L. Li, Scientific Reports 2016, 6, 34367.
[16] aS. Sharma, U. Paiphansiri, V. Hombach, V. Mailander, O. Zimmermann, K. Landfester, V. Rasche, Contrast Media Mol Imaging 2010, 5, 59-69; bA. Servant, I. Jacobs, C. Bussy, C. Fabbro, T. da Ros, E. Pach, B. Ballesteros, M. Prato, K. Nicolay, K. Kostarelos, Carbon 2016, 97, 126-133; cD. Pan, S. D. Caruthers, J. Chen, P. M. Winter, A. SenPan, A. H. Schmieder, S. A. Wickline, G. M. Lanza, Future medicinal chemistry 2010, 2, 471-490; dV. S. Marangoni, O. Neumann, L. Henderson, C. C. Kaffes, H. Zhang, R. Zhang, S. Bishnoi, C. Ayala-Orozco, V. Zucolotto, J. A. Bankson, P. Nordlander, N. J. Halas, Proceedings of the National Academy of Sciences of the United States of America 2017, 114, 6960-6965.
[17] aZ. Cai, C. J. Anderson, Journal of labelled compounds & radiopharmaceuticals 2014, 57, 224-230; bH. D. Lu, L. Z. Wang, B. K. Wilson, S. A. McManus, J. Jumai'an, P. K. Padakanti, A. Alavi, R. H. Mach, R. K. Prud'homme, ACS applied materials & interfaces 2018, 10, 3191-3199.
[18] aJ. Spadavecchia, E. Apchain, M. Alberic, E. Fontan, I. Reiche, Angew Chem Int Ed Engl 2014, 53, 8363-8366; bJ. Politi, L. De Stefano, I. Rea, A. M. Gravagnuolo, P. Giardina, C. Methivier, S. Casale, J. Spadavecchia, Nanotechnology 2016, 27, 195701; cJ. Spadavecchia, D. Movia, C. Moore, C. M. Maguire, H. Moustaoui, S. Casale, Y. Volkov, A. Prina-Mello, Int J Nanomedicine 2016, 11, 791-822; dA. Sette, J. Spadavecchia, J. Landoulsi, S. Casale, B. Haye, O. Crociani, A. Arcangeli, Journal of Nanoparticle Research 2013, 15, 2111.
[19] aN. D’Amelio, C. Esteban, A. Coslovi, L. Feruglio, F. Uggeri, M. Villegas, J. Benegas, S. Paoletti, I. Donati, The Journal of Physical Chemistry B 2013, 117, 13578-13587; bM. A. Mohammed, J. T. M. Syeda, K. M. Wasan, E. K. Wasan, Pharmaceutics 2017, 9, 53; cJ.-Z. Wu, G. R. Williams, H.-Y. Li, D. Wang, H. Wu, S.-D. Li, L.-M. Zhu, International Journal of Nanomedicine 2017, 12, 4037-4057.
[20] S. Kumar, V. K. Meena, P. P. Hazari, S. K. Sharma, R. K. Sharma, Eur J Pharm Sci 2018, 117, 362-370.
[21] J. W. Fredy, J. Scelle, G. Ramniceanu, B. T. Doan, C. S. Bonnet, E. Toth, M. Menand, M. Sollogoub, G. Vives, B. Hasenknopf, Org Lett 2017, 19, 1136-1139.
[22] aG. Kresse, J. Furthmuller, Physical Review B 1996, 54, 11169-11186; bG. Kresse, D. Joubert, Physical Review B 1999, 59, 1758-1775.
[23] aJ. P. Perdew, K. Burke, M. Ernzerhof, Physical Review Letters 1996, 77, 3865-3868; bJ. P. Perdew, K. Burke, M. Ernzerhof, Physical Review Letters 1997, 78, 1396-1396.
[24] aF. Tielens, C. Gervais, J. F. Lambert, F. Mauri, D. Costa, Chemistry of Materials 2008, 20, 3336-3344; bM. Calatayud, F. Tielens, F. De Proft, Chemical Physics Letters 2008, 456, 59-63; cT. V. de Bocarme, T. D. Chau, F. Tielens, J. Andres, P. Gaspard, R. L. C. Wang, H. J. Kreuzer, N. Kruse, Journal of Chemical Physics 2006, 125; dF. Tielens, J. Andres, T. D. Chau, T. V. de Bocarme, N. Kruse, P. Geerlings, Chemical Physics Letters 2006, 421, 433-438.
[25] S. Grimme, J. Antony, S. Ehrlich, H. Krieg, Journal of Chemical Physics 2010, 132.
[26] K. Mathew, R. Sundararaman, K. Letchworth-Weaver, T. A. Arias, R. G. Hennig, Journal of Chemical Physics 2014, 140.
[27] N. H. Ly, T. D. Nguyen, K. D. Zoh, S. W. Joo, Sensors (Basel, Switzerland) 2017, 17.
[28] aF. P. Emmenegger, C. Rohrbasser, C. W. Schläpfer, Inorganic and Nuclear Chemistry Letters 1976, 12, 127-131; bU. Pal, J. F. Sanchez Ramirez, H. B. Liu, A. Medina, J. A. Ascencio, Applied Physics A 2004, 79, 79-84; cM. T. Nguyen, H. Zhang, L. Deng, T. Tokunaga, T. Yonezawa, Langmuir : the ACS journal of surfaces and colloids 2017, 33, 12389-12397.
[29] S. Link, M. A. El-Sayed, The Journal of Physical Chemistry B 1999, 103, 8410-8426.
[30] J. M. Thomas, B. F. G. Johnson, R. Raja, G. Sankar, P. A. Midgley, Accounts of Chemical Research 2003, 36, 20-30.
[31] C. Alric, J. Taleb, G. Le Duc, C. Mandon, C. Billotey, A. Le Meur-Herland, T. Brochard, F. Vocanson, M. Janier, P. Perriat, S. Roux, O. Tillement, Journal of the American Chemical Society 2008, 130, 5908-5915.
[32] aH. Qin, T. Zhou, S. Yang, Q. Chen, D. Xing, Nanomedicine (London, England) 2013, 8, 1611-1624; bT. Guo, Y. Lin, Z. Li, S. Chen, G. Huang, H. Lin, J. Wang, G. Liu, H. H. Yang, Nanoscale 2017, 9, 56-61.
[33] Y. Song, X. Xu, K. W. MacRenaris, X.-Q. Zhang, C. A. Mirkin, T. J. Meade, Angewandte Chemie (International ed. in English) 2009, 48, 9143-9147.
[34] S. G. Boyes, B. Akgun, W. J. Brittain, M. D. Foster, Macromolecules 2003, 36, 9539-9548.
[35] M. D. Rowe, C. C. Chang, D. H. Thamm, S. L. Kraft, J. F. Harmon, Jr., A. P. Vogt, B. S. Sumerlin, S. G. Boyes, Langmuir 2009, 25, 9487-9499.
[36] C. Tian, L. Zhu, F. Lin, S. G. Boyes, ACS Applied Materials & Interfaces 2015, 7, 17765-17775.
[37] J. Spadavecchia, S. Casale, J. Landoulsi, C.-M. Pradier, Chemical Physics Letters 2014, 609, 134-141.
[38] Q. Liu, P. Sacco, E. Marsich, F. Furlani, C. Arib, N. Djaker, M. Lamy de la Chapelle, I. Donati, J. Spadavecchia, Bioconjugate chemistry 2018.
[39] G. Marguerit, H. Moustaoui, M. B. Haddada, N. Djaker, M. L. Chapelle, J. Spadavecchia, Particle & Particle Systems Characterization 2018, 35, 1700299.
[40] H. Liu, P. Jiang, Z. Li, X. Li, N. Djaker, J. Spadavecchia, Particle & Particle Systems Characterization 2018, 35, 1800082.
[41] H. Moustaoui, D. Movia, N. Dupont, N. Bouchemal, S. Casale, N. Djaker, P. Savarin, A. Prina-Mello, M. L. de la Chapelle, J. Spadavecchia, ACS Appl Mater Interfaces 2016, 8, 19946-19957.
[42] J. Politi, L. De Stefano, S. Longobardi, P. Giardina, I. Rea, C. Methivier, C. M. Pradier, S. Casale, J. Spadavecchia, Colloids and surfaces. B, Biointerfaces 2015, 136, 214-221.
[43] H. Moustaoui, D. Movia, N. Dupont, N. Bouchemal, S. Casale, N. Djaker, P. Savarin, A. Prina-Mello, M. L. de la Chapelle, J. Spadavecchia, ACS applied materials & interfaces 2016, 8, 19946-19957.
[44] J. P. López-Neira, J. M. Galicia-Hernández, A. Reyes-Coronado, E. Pérez, F. Castillo-Rivera, The Journal of Physical Chemistry A 2015, 119, 4127-4135.
[45] C. Gullekson, L. Lucas, K. Hewitt, L. Kreplak, Biophysical Journal 2011, 100, 1837-1845.
[46] aW.-H. Park, Z. H. Kim, Nano Letters 2010, 10, 4040-4048; bL. M. Freeman, L. Pang, Y. Fainman, ACS nano 2014, 8, 8383-8391.
[47] R. Juneja, I. Roy, RSC Advances 2014, 4, 44472-44479.
[48] A. Wojtaszek, I. Sobczak, M. Ziolek, F. Tielens, Journal of Physical Chemistry C 2009, 113, 13855-13859.
[49] S. Damoun, W. Langenaeker, G. Vandewoude, P. Geerlings, Journal of Physical Chemistry 1995, 99, 12151-12157.
[50] D. Manna, T. K. Ghanty, Physical Chemistry Chemical Physics 2012, 14, 11060-11069.
[51] G. Ramniceanu, B. T. Doan, C. Vezignol, A. Graillot, C. Loubat, N. Mignet, J. F. Berret, RSC Advances 2016, 6, 63788-63800.

 with y: 1 or 2.
with y: 1 or 2.