A Retrospective Analysis of the Prevalence and Surgical Importance of Hepatic Vascular Variations Detected by Multi-Dedector Computed Tomography
Esin Cibiroglu1* (0000-0003-2825-5518); Davut Tüney2 (0000-0003-0021-7835)
¹Umraniye Training and Research Hospital, Department of Radiology, Istanbul Turkey
²Marmara University Faculty of Medicine, Department of Radiology, Istanbul Turkey
*Corresponding author
Esin Cibiroglu, Umraniye Training and Research Hospital, Department of Radiology, Istanbul-Turkey
Email: dr.esin.sipahi@gmail.com, datuney@hotmail.com
DOI: 10.55920/JCRMHS.2026.14.001589
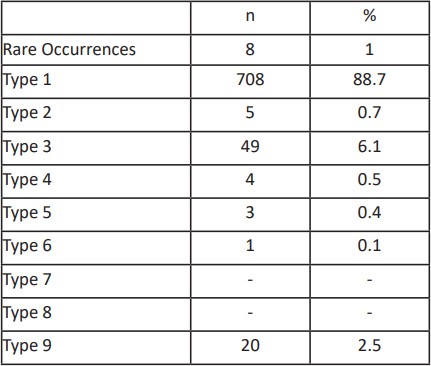
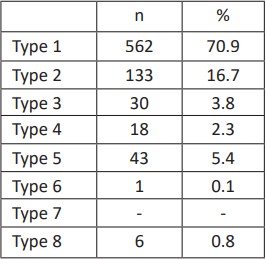
Table 2: Hepatic Venous Variations
Portal Venous Anatomy: In normal anatomy, the main portal vein (PV) is divided into a large right PV (RPV) branch and a smaller left PV (LPV) branch at the level of the liver hilus. The left PV runs horizontally in the medial ligamentum teres. The main part supplies segments II and III of the liver, the superior and inferior branches supply segment IV, and the caudate branches supply segment I. The right PV is divided into anterior (right APV) and posterior (right PPV) trunk. The branches of the anterior trunk supply segments V and VIII, while the branches of the posterior trunk supply segments VI and VII. Any deviation from this structure is considered an anatomical variation.
Type 1 Normal Classical Branching Pattern
Variations;
- Trifurcation (Type 2)
- Origin of the right portal vein (PV) branch as the first branch of the main portal vein (Type 3)
- Quadrifurcation
- Absence of PV bifurcation
- Intrahepatic clustered branching of the portal vein (total ramification)
- Segment VI portal vein branch originating from the main portal vein
- Segment VIII portal vein branch originating from the main portal vein
- Congenital portocaval shunt
Right PV variations;
Normal classical branching pattern
Variations
- -Separate origin of the segment VI portal vein branch from the right portal vein
- -Separate origin of the segment VII portal vein branch from the right portal vein
- -Separate origin of both segment VI and VII portal vein branches from the right portal vein
- -Segment VII portal vein branch originating from the left portal vein
- Trifurcation of the right posterior portal vein
- Quadrifurcation of the right posterior portal vein
Although studies reporting the frequency of hepatic vascular variations in different populations exist in the literature, there is a need for current data from large series using multi-dedector CT (MDCT) in the Turkish population. The aim of this study is to determine the frequency and types of hepatic vascular variations using MDCT in a large patient series and to emphasize the importance of these variations in surgical planning.
Figure 1: Type 3 hepatic artery variation in the coronal MIP image, where the right hepatic artery (red arrow) originates from the superior mesenteric artery.
Figure 2: Type 2 hepatic artery variation in the coronal MIP image, where the left hepatic artery originates from the left gastric artery.
Portal Venous Variations A total of 810 patients were evaluated for portal vein variation. The findings are seen in Table 4.
Table 4: Distribution of Portal Vein Types
The most common variation observed in Type 1 is the separate origin of the segment VII branch from the right portal vein (RPV). The distribution of the variations observed in type 1 is indicated in Table 5 and examples of variations of the portal vein are seen in figures 3, 4 and 5.
Table 5: Distributions of the Variations Observed in Type 1
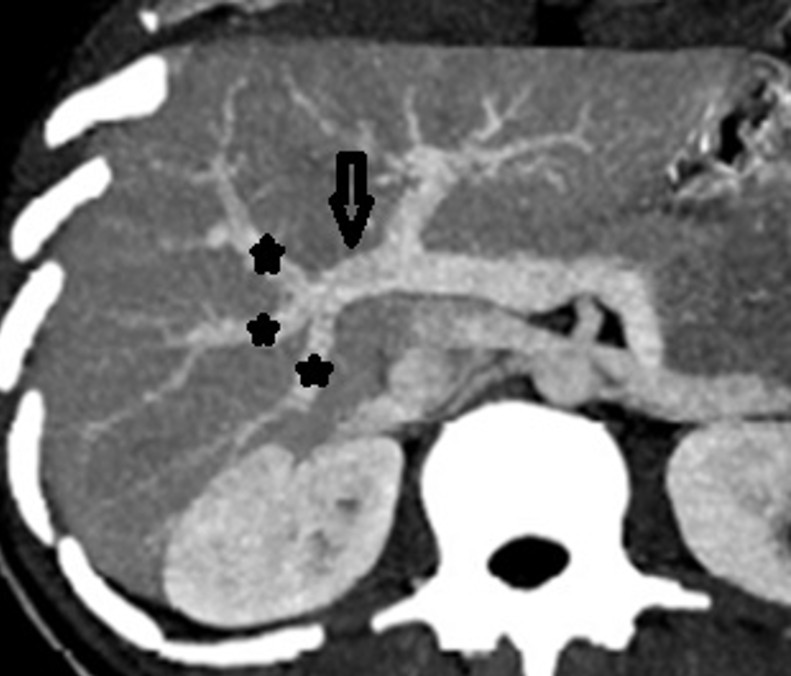
Figure 3: Conventional portal vein anatomy in axial MIP image. The portal vein branches into the right portal vein and the left portal vein. The right portal vein divides into the anterior sector branch supplying segments V and VIII and the posterior sector branch supplying segments VI and VII. The left portal vein divides into branches supplying the left lobe.
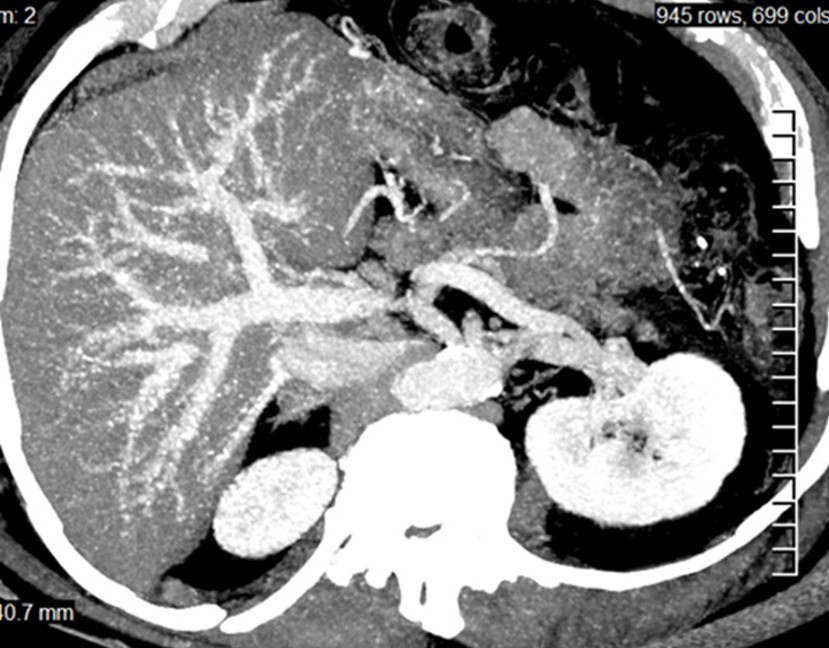
Figure 4: Trifurcation variation in the portal vein on MIP image. Three branches (stars) extend from the main portal vein (arrow): the posterior sector branch supplying segments VI and VII, the anterior sector branch supplying segments V and VIII, and the left portal vein supplying the left lobe.
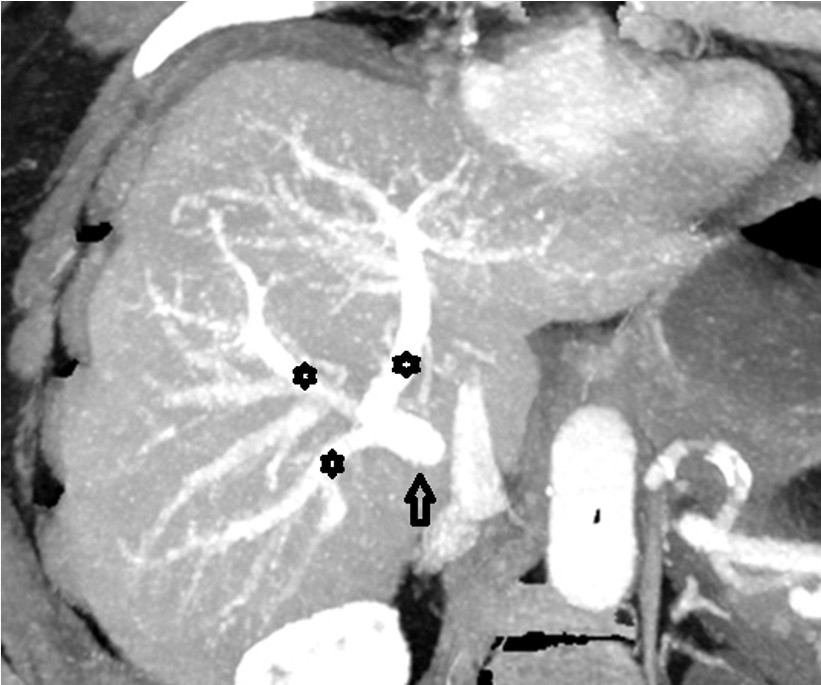
Figure 5: Trifurcation-like branching pattern (stars) in the right portal vein (arrow) on the axial MIP image.
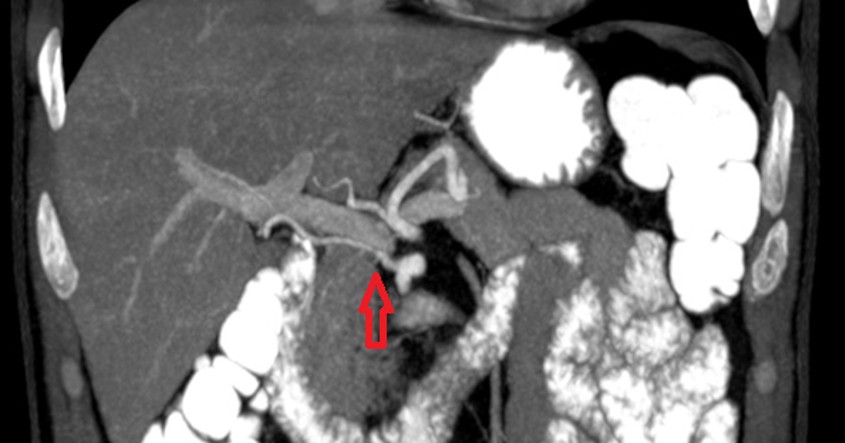
Hepatic Venous Variations 793 patients were evaluated for hepatic vein variation. The findings are seen in Table 6.
Table 6: Distribution of Hepatic Vein Types
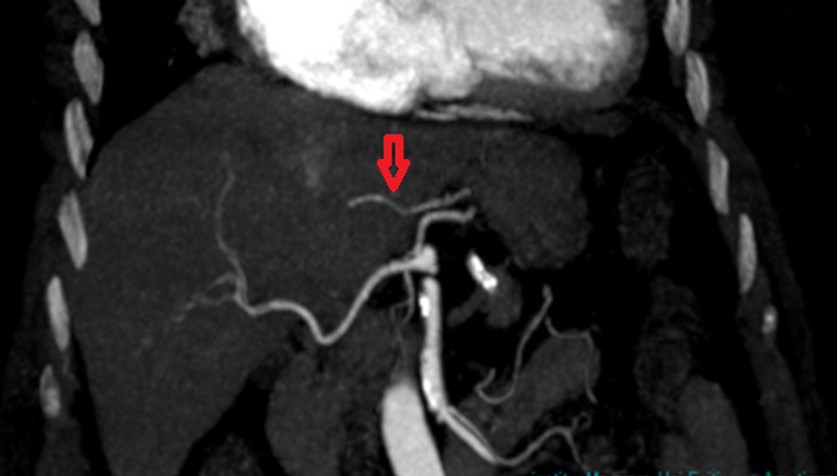
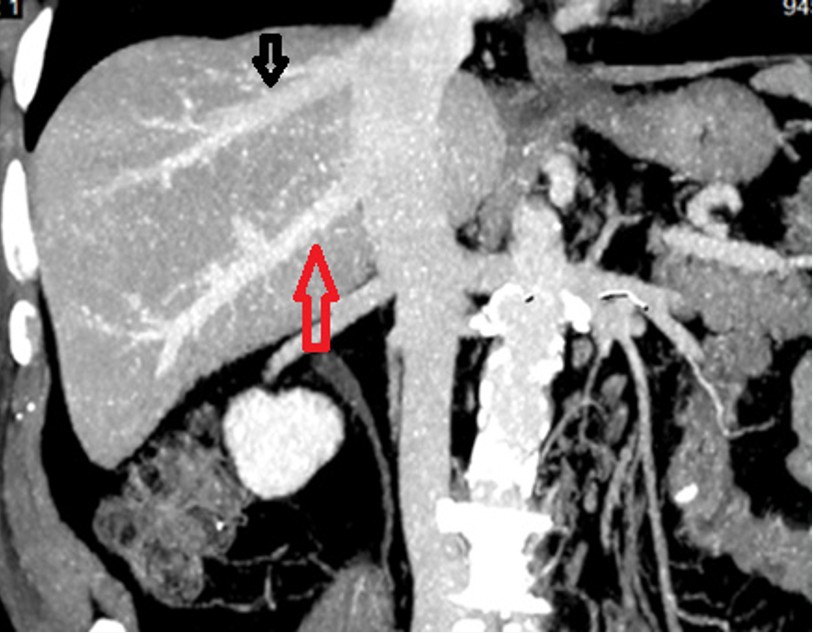
Figure 6: Coronal MIP image. Accessory inferior hepatic vein variation. The accessory right inferior hepatic vein (red arrow), which is almost the same width as the right hepatic vein (black arrow), is seen to open into the inferior vena cava more caudally than the right hepatic vein.