Clinical evidence on the Role of Micronutrients in Male Infertility
Arun M Bourah1, Kedarnath Padte2, Dibyendu Banerjee3 Ashutosh Gautam4, Rajat Singal5
¹Arun M Bourah, Apollo Fertility Guwahati
²Kedarnath Padte, Goa IVF Centre, Panji, Goa
³Dibyendu Banerjee, Ankuran Pluro Fertility, Kolkata, West Bengal
⁴Assistant group lead, Medical Affairs, Mankind Pharma Ltd, Okhla Industrial Estate Phase III, New Delhi, India
⁵General Manager, Medical Affairs, Mankind Pharma Ltd, Okhla Industrial Estate Phase III, New Delhi, India
*Corresponding author
Ashutosh Gautam, Assistant Group Lead I Medical Affairs, Mankind Pharma Ltd., 262, Okhla Industrial Estate
Phase III, New Delhi – 20, India.
Email:ashutosh.gautam@mankindpharma.com
DOI: 10.55920/JCRMHS.2025.13.001587
Abstract
Dietary supplements containing Myo-inositol, N-acetyl cysteine, L-Carnitine, L-Arginine, Selenium, Folic acid, and Zinc are indicated for men with fertility challenges, particularly idiopathic oligoasthenoteratozoospermia (OAT) and male infertility1, 2, 10,11. Male factors contribute to a significant portion of global infertility cases5, ranging from 20% to 40%. Key risk factors include genetic issues, hormonal imbalances, chronic infections, and lifestyle choices such as obesity and substance use5.
A central mechanism in male infertility is oxidative stress (OS), an imbalance between reactive oxygen species (ROS) and antioxidant defences, which damages sperm membranes via lipid peroxidation (LPO) and compromises genetic integrity via DNA fragmentation. The supplement’s ingredients serve as antioxidants or precursors to enhance the body's natural defence mechanisms.
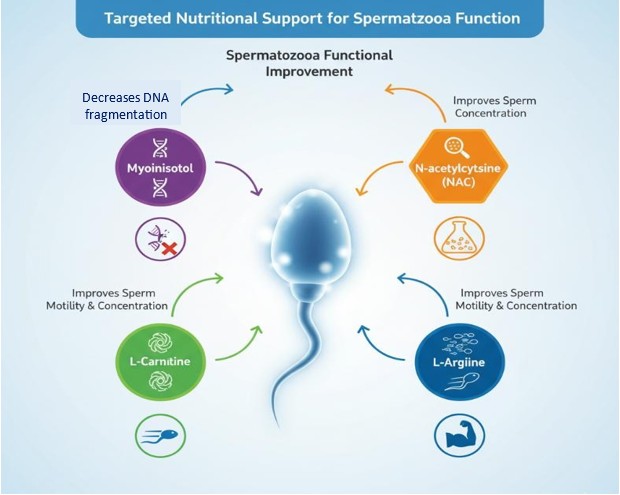
Meta-analyses show that Myo-inositol (MI) significantly improves total and progressive sperm motility and testosterone levels while decreasing sperm DNA fragmentation1,4. N-acetylcysteine (NAC), a glutathione (GSH) precursor, has demonstrated considerable improvement in sperm concentration, ejaculate volume, total sperm motility, and normal morphology2,3. Other components, such as L-Carnitine, Selenium, Zinc, and Folic acid, also play vital roles in sperm metabolism, protection, and DNA integrity.
Infertility, typically defined as the inability to achieve pregnancy after one year of regular, unprotected intercourse, affects approximately 15% of married couples worldwide4. While the experience is an emotional journey shared by many, a significant portion of cases, ranging from 20% to 40%, can be attributed, at least in part, to male factors8. These male factors are often identified through findings related to sperm concentration, motility, or morphology.
A World Health Organization (WHO) multicentric study indicated that 20% of infertility cases were attributed solely to the male partner. Furthermore, data from India suggests that 50% of infertility cases result from reproductive diseases in men. Addressing these male fertility challenges necessitates interventions that can improve the qualitative and quantitative parameters of sperm.8
The key mechanisms of OS-induced sperm damage are:
Lipid Peroxidation (LPO): ROS attack the polyunsaturated fatty acids in the sperm membrane, reducing membrane fluidity, and impairing motility and the sperm's ability to fuse with the egg.
DNA Fragmentation: ROS break DNA strands in sperm cells, compromising genetic integrity and potentially leading to infertility and poor embryo development.
Protein Oxidation: Alteration of structural and functional proteins, which negatively affects motility and fertilization capability.
Myoinositol, N-acetylcysteine and L-carnitine can be particularly indicated in conditions such as idiopathic OAT, male infertility and related fertility challenges.
Keywords: Myo-inositol (MI), Male Infertility, Sperm Motility, Oxidative Stress, Nacetylcysteine (NAC)
Introduction
Infertility, defined as the inability to achieve pregnancy after one year of regular, unprotected intercourse, affects approximately 15% of married couples globally1. Male factors contribute significantly to this challenge, accounting for 20% to 40% of cases worldwide2. Male infertility involves findings related to sperm concentration, motility, or morphology3.
Globally, around 60–80 million couples experience infertility, with prevalence varying by region4. A World Health Organization (WHO) multicentric study found that 20% of infertility cases were attributed solely to the male partner 5, and data from India suggests that 50% of cases result from reproductive diseases in men6. Addressing these challenges necessitates therapeutic interventions that can improve the qualitative and quantitative parameters of sperm7.
The dietary supplements containing micronutrients are designed to enhance male reproductive health by promoting normal spermatogenesis and improving sperm quality and quantity8.
Adjuvants include myo-inositol, N-acetylcysteine, L-carnitine, L-arginine, Selenium, Folic acid, and Zinc. This supplement is particularly indicated for conditions such as idiopathic oligoasthenoteratozoospermia (OAT), male infertility, and related fertility challenges.
Etiology and Risk Factors of Male Infertility
Male infertility is a multifactorial condition driven by both underlying causes and environmental/lifestyle risk factors.
Common Aetiologic Factors
The identified causes of infertility often include:
- Genetic factors.
- Hormonal imbalance.
- Chronic infections
- Lifestyle choices
- Cancer treatments (chemotherapy and radiotherapy).
Significant Risk Factors
A number of factors increase the risk of male infertility1:
- Obesity/Overweight: Body mass index (BMI) greater than 251.
- Age: 40 years or above1.
- Toxin Exposure: Exposure to environmental toxins such as Lead and Mercury2.
- Substance Use: Use of Tobacco, Alcohol, and Marijuana2.
- Medications: Use of Anabolic Steroids 8, Depo preparations of testosterone 8, androgen receptor blockers 8, and oestrogenic drugs8.
- Medical History: History of post-pubertal mumps infection 8 or Chronic Liver Disease8.
- Anatomical Issues: Presence of varicocele 8or unilateral/bilateral undescended testes8.
Obesity and Metabolic Disruption
Obesity significantly compromises reproductive health by disrupting the hypothalamicpituitary-gonadal (HPG) axis, which leads to hormonal imbalances and impaired semen parameters7. Obesity-mediated damage includes physical factors like increased scrotal temperature from heavy adipose tissue, as well as systemic inflammation and oxidative stress (OS)7.
Obesity is also a primary driver of secondary hypogonadism in moderate to severely obese males, with a prevalence of about 45%7. This condition is marked by hormonal imbalances, including decreased testosterone and SHBG, and increased estrogen levels7. The inverse relationship between BMI and plasma testosterone concentration is partly due to increased intraadipocyte aromatase activity, which converts testosterone to 17𝛽-estradiol. This leads to a complex "hypogonadal-obesity cycle" that further lowers testosterone and increases fat deposition.
Furthermore, Type 2 Diabetes (T2DM) and insulin resistance are strongly interlinked with obesity and defective spermatogenesis35. Hyperinsulinemia inhibits normal spermatogenesis, and diabetic males show higher nuclear and mitochondrial DNA damage in sperm3. Hypertension, a major cardiovascular risk factor, also shows a significant inverse relationship with total serum testosterone and free testosterone levels7.
The Central Role of Oxidative Stress in Sperm Dysfunction: Oxidative stress (OS) is a major contributor to sperm dysfunction and male infertility, resulting from an imbalance where excessive
Reactive Oxygen Species (ROS) production overwhelms the body’s antioxidant defences.
Mechanisms of OS-Induced Damage: While low levels of ROS are beneficial for normal sperm function, high levels are harmful. ROS attack sperm through several primary mechanisms4:
- Lipid Peroxidation (LPO): ROS attack the polyunsaturated fatty acids (PUFAs) in sperm membranes, reducing fluidity and impairing motility and fusion capability4,1.
- DNA Fragmentation: ROS break DNA strands, compromising genetic integrity and potentially causing poor embryo development4,2.
- Protein Oxidation: Alters structural and functional proteins, negatively affecting motility and fertilization capacity4,3.
Spermatozoa are highly susceptible to OS because they lack cytoplasmic antioxidant repair systems and possess membranes rich in PUFAs4. Elevated ROS levels are found in 25%–40% of infertile men compared to fertile men, and this OS negatively correlates with semen quality and pregnancy rates4,5. This makes reversing OS a critical therapeutic strategy in infertility treatment4,6.
Role of Specific Micronutrients in Male Infertility: Antioxidant use in patients with idiopathic OAT mainly relies on the rationale of protection that each antioxidant provides against OS. The following antioxidants were chosen based on available evidence supporting spermatogenesis, shielding against OS, and enhancing semen parameters.
Myo-inositol (MI): MI is the most prominent of nine inositol stereoisomers and participates in numerous biological processes4. In the male reproductive tract, MI and its derivatives maintain endoplasmic reticulum (ER) stress control in the testis and epididymis4. MI is critical for:
- Spermatogenesis and Maturation: Contributes to the degradation of misfolded proteins and is essential as spermatozoa mature in the epididymis, helping them acquire progressive motility5.
- Sperm Function: MI phosphoderivatives regulate hyperactivated motility, thermotaxis (guidance), actin cytoskeleton remodeling, and the acrosome reaction5.
- Clinical Evidence: A systematic review and meta-analysis of 16 selected studies found significant improvements in several sperm parameters after MI administration5.
Total Sperm Motility: Showed a significant increase (SMD 0.90; p=.001)5.
Progressive Sperm Motility: Showed a significant increase (SMD 1.48; p=.008)5.
Testosterone Level: Showed a significant improvement (SMD 0.54; p<.0001)5.
DNA Fragmentation: Showed a significant decrease (SMD −1.37; p=.01)5.
Pregnancy Rate: The pooled pregnancy rate in MI-receiving groups was 34% (95% CI: 21% to 48%)5.
N-acetylcysteine (NAC): NAC is a potent antioxidant widely used as a mucolytic agent2. Its primary antioxidant importance is due to its ability to increase levels of intracellular cysteine, which is a precursor to the major intracellular antioxidant Glutathione (GSH)3. GSH is pivotal for glutathione peroxidase (GPx), which neutralizes peroxides and protects sperm membranes from LPO, thus preserving motility3.
The mechanism of action for NAC includes:
- GSH Precursor: Boosts GSH synthesis to neutralize ROS in seminal plasma2.
- Direct Scavenging: Has a free thiol group that directly scavenges ROS2.
- Reduced Damage: Decreases malondialdehyde (MDA), a marker of LPO2.
- Clinical Evidence: Meta-analyses of randomized controlled trials (RCTs) demonstrated that NAC therapy resulted in significant improvements for idiopathic infertile men:
- Sperm Concentration: Mean difference (MD) of 3.80 (p<0.00001) in one analysis3, and MD of 4.43 (p=0.003) in another3.
- Total Sperm Motility: MD of 9.69 (p<0.00001)3.
- Normal Morphology: MD of 1.36 (p<0.0001)3.
- Ejaculate Volume: MD of 0.69 (p=0.002)3.
- NAC at 600mg for 12 or 26 weeks improved sperm quality with no reported side effects3.
L-Carnitine and L-Arginine
- L-Carnitine (LC): A water-soluble antioxidant that plays a crucial role in sperm metabolism and motility9. Men with OAT have significantly lower levels of carnitine in their semen9. An RCT meta-analysis showed that LC monotherapy significantly increased sperm concentration, improved normal morphology, and enhanced sperm motility9.
- L-Arginine: A semi-essential amino acid and a precursor to nitric oxide (NO), a molecule critical for vascular function, sperm motility, and erectile physiology10. L-Arginine enhances NO synthesis, improving blood flow to the testes and supporting spermatogenesis74. Supplementation can increase sperm motility and concentration, particularly in men with OAT10. L-Arginine often shows synergistic effects when combined with other nutrients10.
Selenium: Selenium is an essential trace element involved in spermatogenesis and protects sperm DNA from OS11,10. It functions through selenoproteins, such as GPX enzymes, which maintain sperm structural integrity78. Deficiency is linked to midpiece abnormalities and impaired sperm motility10,11. Treatment with 200µg of Selenium, alone or with NAC, has been reported to significantly improve all semen parameters11.
Zinc: Zinc is essential for the metabolism of DNA and RNA, supporting cellular function and spermatogenesis10,11. It acts as a powerful antioxidant and has antiapoptotic effects, protecting sperm cells from oxidative damage10. Seminal plasma zinc levels are significantly higher in fertile men11. Deficiency is linked to defects in sperm structural integrity, leading to flagellar abnormalities, axonemal disruption, and abnormal or absent midpiece10.
Folic Acid: Folic acid is vital for DNA/RNA synthesis and amino acid metabolism, supporting cellular and reproductive functions11. It acts as a free radical scavenger. Folic acid intake is linked to a lower frequency of Sperm DNA Fragmentation (SDF), and daily intake of 700 µg is associated with a 30% lower risk of chromosomal abnormalities like disomy X and sex nullisomy10, 11.
Discussion
There is a steady rise in the male component of infertility cases. It is primarily influenced by faulty spermatogenesis happening in the testicles. These spermatozoa might be deficient in multiple parameters, like total absence (azoospermia), low count (Oligospermia), and abnormal morphology (teratozoospermia). The success in dealing with male infertility is limited because of a multifaceted aetiology, such as that which arises from the interaction of genetics, lifestyle choices, environmental influences, and concomitant conditions. Research indicates that reactive oxygen species contribute to sperm damage in 30-80% of cases12. GSH is the primary nonprotein thiol in the body and a key antioxidant that maintains cellular redox status. It directly reacts with reactive species and serves as a cofactor for antioxidant enzymes. Since cysteine levels are lower intracellularly, this limits GSH biosynthesis, highlighting NAC's role as a prodrug for cysteine and GSH. Neither GSH nor cysteine increased cellular GSH levels, underscoring NAC as a major strategy to reduce oxidative stress.
MI and its synthesizing enzymes are involved in spermatogenesis, spermiogenesis, maturation, and development of male gametes in the seminiferous tubules of the testis. The physiologic concentration of MI is also associated with balanced levels and normal functions of reproductive hormones, including FSH, inhibin-B, insulin, and TSH.
Sperm are exposed to increasing MI levels during their migration from the epididymis toward the deferent duct, which is synthesized by the male reproductive organs or received from blood circulation. This factor plays a role in sperm maturation and function by producing inositol triphosphate and opening calcium channels. MI is essential in many signaling pathways responsible for regulating cytoplasmic calcium levels, capacitation, mitochondrial function, and the frequency of tail beating6.
NAC and MI supplementation is probably beneficial to increase the chance of natural or assisted pregnancy in infertile couples by improving semen parameters.
Conclusions
Male factor infertility is a significant global health issue, contributing to as much as 40–50% of cases8. The available literature strongly indicates that the combination of Myo-inositol, N-acetylcysteine, L-carnitine, L-arginine, Zinc, Selenium, and Folic acid can significantly improve sperm parameters by directly enhancing antioxidant defences and supporting the complex processes of spermatogenesis and sperm function.
By addressing the core pathological mechanism of oxidative stress and mitigating associated risks such as metabolic dysfunction, interventions like Myoinositol and N-acetylcysteine serve as an essential dietary strategy. Systematic reviews support that these antioxidants not only enhance sperm quality but also increase pregnancy rates.
References
- Ghaemi M, Seighali N, Shafiee A, Beiky M, Kohandel Gargari O, Azarboo A, et al. The effect of Myo-inositol on improving sperm quality and IVF outcomes: A systematic review and metaanalysis. Food Sci Nutr. 2024 Sep 12;12(11):8515–24.
- Tenório MCDS, Graciliano NG, Moura FA, Oliveira ACM, Goulart MOF. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants (Basel). 2021 Jun 16;10(6):967.
- Zhou Z, Cui Y, Zhang X, Zhang Y. The role of N-acetyl-cysteine (NAC) orally daily on the sperm parameters and serum hormones in idiopathic infertile men: A systematic review and meta-analysis of randomised controlled trials. Andrologia. 2021 Mar;53(2):e13953.
- Vazquez-Levin MH, Verón GL. Myo-inositol in health and disease: its impact on semen parameters and male fertility. Andrology. 2020 Mar;8(2):277–98.
- Kumar S, Agrawal D, Sharma K, Swain TR. Association of Male Infertility to Metabolic Syndrome and Other Related Disorders. J Integr Nephrol Androl. 2015;2(3):107–16.
- Bashiri Z, Sheibak N, Amjadi F, Zandieh Z. The role of myo-inositol supplement in assisted reproductive techniques. Hum Fertil (Camb). 2023;26(5):1044–60.
- Chaudhuri G, Das A, Kesh S, Bhattacharya K, Dutta S, Sengupta P, et al. Obesity and male infertility: multifaceted reproductive disruption. Middle East Fertil Soc J. 2022;27(1):8.
- Priyadarshi S, Tanwar R, Malhotra V, Vinod KV, Krishnappa P, Ali I, et al. The Urological Society of India guidelines for the management of male infertility (Executive Summary). Indian J Urol. 2023 Jan-Mar;39(1):7–11.
- Rastgoo S, Fateh ST, Nikbaf-Shandiz M, Rasaei N, Aali Y, Zamani M, Shiraseb F, Asbaghi O. The effects of L-carnitine supplementation on inflammatory and anti-inflammatory markers in adults: a systematic review and dose–response meta-analysis. Inflammopharmacology.
- 2023;31(6):2173–2199. doi:10.1007/s10787-023-01323-9
- Zhou X, Shi H, Zhu S, Wang H, Sun S. Effects of vitamin E and vitamin C on male infertility: a meta-analysis. Int Urol Nephrol. 2022;54:1793–1805. doi:10.1007/s11255-022-03237-x.
- Agarwal A, Kashou AH, Sekhon LH. Oxidative stress and the use of antioxidants for idiopathic OATs. In: Agarwal A, editor. Studies on Men's Health and Fertility. New York: Springer; 2012. p. 485–516. doi:10.1007/978-1-61779-776-7_22
- Kizildag S, Gürkan T, Gürsoy H, Özer E. Unlocking the potential of antioxidant supplementation with N-acetylcysteine to improve seminal parameters and analysis of its safety: A systematic review and meta-analysis of randomized controlled trials. Reprod Biol Endocrinol. 2024;22(1):42.