Effectiveness of Piracetam in Retinitis Pigmentosa: A Case Report
Lan T.D. Le, MD, PhD1*, Sang T.D. Le, MD2
¹Assosiate Professor, Pham Ngoc Thach University of Medicine, Vietnam
²Ho Chi Minh City Eye Hospital, Vietnam
*Corresponding author
Lan T.D. Le,Assosiate Professor professor Pham Ngoc Thach University of Medicine Vietnam
Email: hoanglelan_bs@yahoo.com, sangledo@gmail.com
DOI: 10.55920/JCRMHS.2025.12.001529
Abstract
Objective: Evaluate the effectiveness of Piracetam in retinitis pigmentosa (RP).
Method: A case report of a clinical trial off label of a patient with retinitis pigmentosa with Piracetam 12g/bottle, 4 courses in 12 months.
Result: A male patient, 29 years old, came for examination because of blindness in both eyes. Binocular vision was hand motion, peripheral vision was completely lost. Clinical examination revealed that the anterior segment was normal, the fundus had spongy cell pigments scattered throughout the peripheral retina, spreading to the macular. The patient was diagnosed with retinitis pigmentosa and received a blinded clinical trial with intravenous Piracetam 12g in several courses, each course lasting 5 - 10 consecutive days. After 4 courses, visual acuity of the right eye was counting fingers (CF) 0.7m and the left eye was CF 1.5m, and beside vision in both eyes was widen to 135 degrees of the temporal side.
Conclusion : Retinitis pigmentosa is a disease, which seriously threatens vision from a young age. Currently, there is no specific treatment for RP. Ongoing therapies, which are mostly based on genes and cells with extremely high price, are only used for groups of patients with specific gene mutations. Treatment with Piracetam in this off label clinical trial initially improved low vision, but the patient was able to do himself living activities basically daily.
Keywords : Retinitis Pigmentosa, Piracetam.
Introduction
Retinitis pigmentosa (RP) is a group of hereditary retinal diseases caused by a collection of different gene mutations, which involve the gradual loss of photoreceptor cells, including rod and cone cells, and loss of pigment epithelial function. The disease usually occurs in both eyes, characterized by progressive vision loss. The first stage is manifested by progressive degeneration and death of rod cells. Therefore, the initial symptom is reduction in night vision or night blindness, and gradually narrow peripheral visual field. In the late stage, cone cells are also affected, leading to progressive vision loss, tunnel vision and color vision defect. Before the age of 40, most patients only see light perception. This results in difficulty at work, increase in anxiety, depression and social isolation; which greatly lower patient's quality of life.
Currently, there are 131 drugs, excluding Piracetam, that have been reported at all stages of clinical trial for the treatment of retinitis pigmentosa (1). Within them, gene therapy and stem cell therapy are two main methods. Presently, FDA only approves two methods, include: (1) gen therapy called Voretigene neparvovec (brand name Luxturna), which is suitable for a small number of patients having RPE65 gene mutation (account for 0.3-1% of all retinitis pigmentosa patients); and (2) The Argus-II retinal prosthesis implantation. These methods have some disadvantages, including extremely high price, the patients must suffer from a vitrectomy and subretinal injection or pre-retinal implant, which require skillful surgeons. Therefore, both of them are still not popular, especially in developing countries.
Piracetam has been shown to be effective in anti-oxidative stress in neurological diseases. On the other hand, previous studies have shown many factors related to the pathogenesis of retinitis pigmentosa, including oxidative stress. Thus, with the aim of helping patients to recover their vision to a certain degree, in order to be able to do personal activities on their owns, we conducted an off-label trial on the treatment of retinitis pigmentosa with Piracetam.
Methods
A 29 year-old man named Hoang Van P., who works as a blind massage therapist and resides in District 10, Ho Chi Minh City, came for examination because he was blind in both eyes.
Medical history: The patient reported that his vision had gradually worsened and been painless since he was in kindergarten, without previous ocular trauma. In 2010, he came to a private clinic for an eye examination. His best corrected visual acuity (BCVA) was 20/200 in the right eye and 20/40 in the left eye. In 2012, his right eye was diagnosed with 40 degrees exotropia and amblyopia. After that, he went to the Vietnam national eye hospital and experienced an electroretinogram (ERG) test. The result was complete retina destruction in both eyes, and he was diagnosed with retinitis pigmentosa (RP) in both eyes. The patient did not receive any treatments. Currently, both eyes are almost blind, and the patient must be guided by others to do daily activities. The patient came to the Eye department of Thanh Quan general clinic, being diagnosed with RP in both eyes. The patient asked for a treatment to help him walk by himself.
Past medical history: He was full-term born without any birth defects. He does not have hearing impairment or any systemic diseases. Family history of retinitis pigmentosa is unknown.
Examination:
Visual acuity (VA): Hand motion OU
Intraocular pressure: 20.6 mmHg OU
Centre vision: tunnel vision OU
Peripheral vision: completely loss OU
Color recognition: the patient only identifies 2 colors including white and black
Microscopic examination: The anterior segment of both eyes is normal.
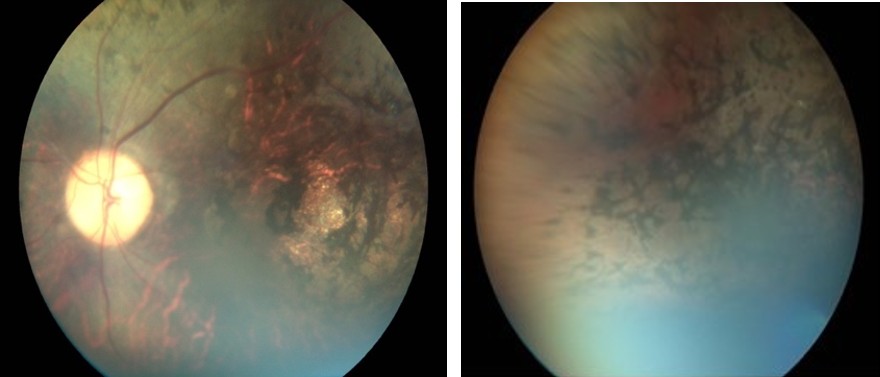
Ophthalmoscopy with indirect ophthalmoscope showed that pigment cells scattered througout the peripheral retina, spreading into the macular; optic disc pallor and contricted arteries.
Fundus photography showed that:
Figure 1 : Patient Hoang Van P.
Figure 2 : Fundus photo of both eyes.
The patient did not perform OCT or OCT-A scan test, ERG test, visual field test, color blind test and fluorescein angiography (FA) test.
Treatment:
The patient was explained that in Vietnam, there is currently no specific treatment for this disease. There are only 2 drugs approved by the FDA, including Luxturna and Argus II implanation. However, the costs of these drugs are extremely high, which are $850,000 and $115,000 - $150,000 respectively. Nevertheless, he still wished for help, in other to improve his vision to a certain degree. Therefore, we explained to him that a drug called Piracetam is effective in supporting some neurological, otorhinolaryngological and ophthalmological diseases, such as ischemic stroke, sudden deafness, ischemic optic neuropathy; but we have never utilized it in RP treatment. Then, we conducted an off label clinical trial to the patient with Piracetam, which was approved by the patient (no images or videos of the patient’s activities before this trial were recorded).
Method: intravenous injection of Piracetam at a dose of 12 grams per day, for 5 - 10 days interval with repetition if required as determined upon examination on the final day of injection within each round.
Results
Course 1 ( March 6th - March 15th, 2023): intravenous injection of Piracetam at a dose of 12 grams per day of 10 days. VA increased to CF 0.2m OD and CF 0.5m OS. The patient was then scheduled for a follow up visit in 10 days.
Course 2 ( March 27th - April 3rd, 2023): intravenous injection of Piracetam at a dose of 12 grams per day of 8 days. VA increased to CF 0.7m OD and CF 1.5m OS. Peripheral vision in both eyes was widen to 135 degrees towards the temporal side. In addition, color vision also improved, the patient could identify 3 color, including white, black and pink. In which, the patient mainly saw white and black colors, and occasionally pink.
After two rounds of treatment, the patient was able to do daily activities on his own, such as walking by himself, seeing a door knob, opening the door, and detecting numbers on a cash. We decided to stop the treatment and made a follow up examination after 6 months.
Course 3 (September 18th - September 22th, 2023): After 6 months, the patient was re-checked at our clinic. At September 18th, 2023: the patient reported that his vision in both eyes gradually decreased during the non-treatment period; however, peripheral vision was not narrow. Examination showed that his VA was CF 0.3m OD and CF 0.5m OS.
Intravenous injection of Piracetam at a dose of 12 grams per day of 5 days. VA increased to CF 0.7m OD and CF 1.5m OS. The patients was discontinued the treatment and was made a follow-up appointment in 6 next months.
Course 4 ( March 18th - March 22th, 2024): After 6 months, the patiend was re-examined. At March 18th, 2024: examination showed that his VA was CF 0.5m OU. Peripheral vision and color vision remained unchanged. The patient was treated a fourth course:
Intravenous injection of Piracetam at a dose of 12 grams per day of 5 days. VA rose to CF 0.7m OD and CF 1m OS.
July 12th, 2024: After 4 months, the patient came back to our clinic. VA was CF 1m OD and CF 1m OS. Peripheral vision and color vision remained unchanged.
During the treatment, side effects of Piracetam such as drug allergy, weight gain, hyperactivity, anxiety, weakness, depression were not recorded; blood tests and liver and kidney functions were within normal limits.
Figure 3 : Patient changes slippers on his own
Figure 4 : Patient sits down by himself
Discussion
Retinitis pigmentosa (RP) is a group of hereditary retinal diseases caused by a collection of different gene mutations, which involve the gradual loss of photoreceptor cells, including rod and cone cells, and loss of pigment epithelial function.
The disease usually occurs in both eyes, relatively symmetrical, having many different clinical manifestations and is divided into two groups: (1) with systemic diseases and (2) without systemic disease, in which simple retinitis pigmentosa accounts for the large number of cases. The fundus has three typical signs: bone-shaped black pigments, constricted blood vessels and optic disc pallor. These bone-shaped black pigments clustered in clumps of cells along or surrounding blood vessels originating from the retina behind the equator, then spreading to the peripheral and central retinal. When Bruch's membrane is broken, the retina gradually thins and blood vessels constrict, especially capillaries and arteries. In the late stage, the optic disc is pale and atrophic. Some paraclinical tests include visual field test, colour vision test, electroretinography, OCT, and fundus fluorescein angiography. Electroretinography is an important test, which is used to assess hereditary retinal diseases, performed early even when the disease does not present any symptoms.
Currently, there is no specific treatment for retinitis pigmentosa. In the 20th century, some countries, including Vietnam, used placenta extracts (Placentil) to treat this disease, but were not successful (2). After that, many trials were conducted with the aim of successfully treating retinitis pigmentosa, mainly gene therapy and stem cell therapy. Currently, there are 131 drugs all over the world being researched, not including Piracetam, which have been reported at all stages of clinical development in the treatment of retinitis pigmentosa.
There are two FDA-approved methods in 2022, include: (1) gene therapy Voretigene neparvovec (Luxturna, USA) and (2) Electronic retinal implant therapy Argus II (Canada). Luxturna is used for patients with RPE65 gene mutations with an extremely expensive price at 850 thousand USD for both eyes; patients need to undergo vitrectomy and subretinal injections, which require skillful surgeons. Studies reported that Luxturna improves visual field and light sensitivity threshold; however, the BCVA does not change (2, 3), and the most common complication that can occur is retinal atrophy. Meanwhile, Argus II is used for patients who can only see light, including a chip system on the retina and a camera on the glasses frame.
Other treatments include stem cells, retinal photocoagulation laser, some drugs and herbs (1,3-5). Current studies show that high dose vitamin A 15,000 units and DHA are not effective in treating RP because they do not change the visual field, electroretinography; in contrast, excess vitamin A affects liver function, causes birth defects, increases lipofuscin accumulation in the retina, which worsen RP. Otherwise, Chen Ou (China) tested on mice, using extracts from Fructus Lycii and Salvia Miltiorrhiza Bunge with the aim of improving retinal morphology and function, by regulating the Nrf2/HO-1 pathway to inhibit oxidative reactions (4). However, the effectiveness of these therapies is still very low, or unsuccessful. The main goal is restoring vision at the level of incomplete blindness, the patient can walk and live independently.
Regarding the pathogenesis of retinitis pigmentosa, previous studies noted that oxidative stress is related to the death of photoreceptor cells in the retina. Rod cells are the most numerous in the retina, accounting for 95% of the outer nuclear layer, containing many mitochondria, having the strongest metabolic activity, as well as consuming the most oxygen. When rod cells die due to genetic mutations, oxygen consumption in the outer retina significantly decreases. Meanwhile, the oxygen supply is not reduced, leading to increased oxygen levels in the outer retina; retinal vasoconstriction shows that oxygen flows from the outer retina to the inner retina (8). Studies conducted on animals showed that an increase in oxygen levels leads to progressive oxidative damage in cone cells. The density of cone cells in the mid-periphery of the retina is low; when rod cells die, cone cells in this area are exposed to a particularly high amount of tissue oxygen levels, leading to the start of cone cell degeneration. When the cone cells in this area die, the clinical feature shows a ring scotoma. The remaining cone cells at the inner and outer edges of the scotoma are exposed to the highest amount of oxygen in the tissue, which can be predicted that the scotoma will spread to the periphery and centre. The process of cone cell death is slow and continuous. Because the density of cone cells in the fovea is the highest, the cone cells here survive the longest. When the cone cells in this area die, it leads to loss of central vision, and the patient becomes completely blind (8).
Piracetam, which is a derivative of the neurotransmitter GABA (Gamma Amino-Butyric Acid), is a substance that improves the metabolism of nerve cells such as Acetylcholine, Noradrenaline, Dopamine, helping these cells function better. Studies conducted on Alzheimer's and Parkinson's patients showed that oxidative stress and loss of mitochondrial function are one of the main causes of nerve cell death. Mitochondria are one of the main sources of oxidative stress due to the use of oxygen for energy, related to caspase protein-dependent and -independent pathways. Oxidative stress can cause neuronal death through a caspase-independent pathway. Researchers showed that Piracetam produces anti-inflammatory, antioxidant effects and prevents cell death. Oxidative stress increases the concentration of reactive oxygen species (ROS), which is inhibited by treatment with Piracetam. Piracetam improves mitochondrial function by increasing the fluidity of the mitochondrial membrane (6). Currently, the treatment of neurological and ophthalmic diseases with Piracetam has not been approved by the FDA because of some side effects such as increased movement, weight gain, anxiety, drowsiness, depression, weakness, decreased platelet aggregation as well as fibrinogen concentration, and is contraindicated in patients with cerebral haemorrhage and renal failure (7).
In our study, treatment of RP with Piracetam was effective in improving central vision, peripheral vision and colour perception. This recovery is largely due to the antioxidant effect of Piracetam on the oxidative stress process in the retina. When exposed to high oxygen levels in the retina for a long time, cone cells gradually die; this is improved by the inhibitory effect of Piracetam on oxidative stress, which prolongs the life of cone cells. Because the patient came to the clinic late, with a significant decrease in his vision, we thought that a large number of rod cells and cone cells died. Therefore, treatment with Piracetam is only effective in preventing the oxidative stress process occurring in the remaining cone cells, mainly in the macula. On the other hand, because oxidative stress in the retinal tissue is continuous; thus, when treatment with Piracetam is stopped, the cone cells continue to be exposed to high oxygen levels. This requires constant treatment with Piracetam. After 4 courses of treatment with Piracetam, the patient currently has no side effects and liver and kidney function tests are within normal limits. However, because long-term treatment is required, it is necessary to closely monitor the side effects of Piracetam on patients. Our study was conducted as an off label trial, so the diagnosis and monitoring are mainly based on clinical practice. Therefore, monitoring of structural and functional changes after treatment with Piracetam based on paraclinical studies is still lacking. Our study provides initial assessments of the effectiveness of Piracetam in the treatment of retinitis pigmentosa, creating the premise for following studies that will perform in different clinical conditions, with sufficient paraclinical tests taken during and after treatment, to demonstrate the effectiveness of Piracetam.
Conclusion
This report summarizes a case of retinitis pigmentosa treated with Piracetam. This is a genetic disease and is not rare.
The disease appears early from young age, causes progressive vision loss. The ultimate consequence is vision loss, negatively affecting the patient’s quality of life. Currently, there is no specific treatment. The only two treatments approved by the FDA are Luxturna and Argus II, only applicable to a small group of RP patients and the costs are extremely high. Thus, these treatments are not unpopular widespread.
Off label trial treating RP with Piracetam helps to improve vision to a certain degree, and the patient was able to do daily activities on his own.
Patient consent: Consent to publish the case report was not obtained. This report does not contain any personal information that could lead to the identification of the patient.
Funding: No funding or grant support
References
- Cross N, van Steen C, Zegaoui Y, Satherley A, Angelillo L. Current and Future Treatment of Retinitis Pigmentosa. Clin Ophthalmol. 2022;16:2909-21.
- Das Gupta BK, Sen GC. Tissue Therapy - An analysis of the result obtained by the use of aqueous extract of human placenta in 50 unselected cases. Indian Journal of Ophthalmology. 1953;1(2):57-60.
- Ou C, Jiang P, Tian Y, Yao Z, Yang Y, Peng J, et al. Fructus Lycii and Salvia miltiorrhiza Bunge extract alleviate retinitis pigmentosa through Nrf2/HO-1 signaling pathway. J Ethnopharmacol. 2021;273:113993.
- Kang S, Lorach H, Bhuckory MB, Quan Y, Dalal R, Palanker D. Retinal Laser Therapy Preserves Photoreceptors in a Rodent Model of MERTK-Related Retinitis Pigmentosa. Transl Vis Sci Technol. 2019;8(4):19.
- Farvardin M, Afarid M, Attarzadeh A, Johari MK, Mehryar M, Nowroozzadeh MH, et al. The Argus-II Retinal Prosthesis Implantation; From the Global to Local Successful Experience. Front Neurosci. 2018;12:584.
- Verma DK, Gupta S, Biswas J, Joshi N, Singh A, Gupta P, et al. New therapeutic activity of metabolic enhancer piracetam in treatment of neurodegenerative disease: Participation of caspase independent death factors, oxidative stress, inflammatory responses and apoptosis. Biochim Biophys Acta Mol Basis Dis. 2018;1864(6 Pt A):2078-96.
- Cohen PA, Zakharevich I, Gerona R. Presence of Piracetam in Cognitive Enhancement Dietary Supplements. JAMA Intern Med. 2020;180(3):458-9.
- Yu DY, Cringle SJ, Su EN, Yu PK. Intraretinal oxygen levels before and after photoreceptor loss in the RCS rat. Invest Ophthalmol Vis Sci. 2000;41(12):3999-4006.