Introduction
Tuberculosis, caused by the bacterium Mycobacterium tuberculosis (Mtb), remains one of the most significant infectious diseases globally, affecting millions of people each year. Despite substantial advancements in medical science, TB continues to be a leading cause of morbidity and mortality worldwide. The World Health Organization (WHO) estimates that in 2021 alone, approximately 10.6 million people fell ill with TB, and 1.6 million succumbed to the disease, making it one of the top ten causes of death globally(1). TB is particularly prevalent in low- and middle-income countries, where access to healthcare and treatment is often limited(2). The disease primarily affects adults in their most productive years, leading to significant economic losses and burdening healthcare systems. Additionally, TB has a close association with poverty, malnutrition, and other conditions that weaken the immune system, such as HIV/AIDS(3). The interplay between TB and HIV is particularly concerning, as co-infection dramatically increases the risk of active TB and complicates treatment outcomes(4). One of the most formidable challenges in the fight against TB is the emergence and spread of drug-resistant strains of Mycobacterium tuberculosis. Drug-resistant TB occurs when the bacterium develops resistance to one or more of the standard anti-TB drugs. The most severe form is multidrug-resistant TB, defined as resistance to at least isoniazid and rifampicin, the two most potent first-line anti-TB drugs(5). Even more alarming is extensively drug-resistant TB, which is resistant to isoniazid and rifampicin, in addition to any fluoroquinolone and at least one of the second-line injectable drugs (amikacin, kanamycin, or capreomycin)(6). The treatment of MDR-TB and XDR-TB is significantly more complex, costly, and prolonged, often with severe side effects and lower success rates. The rise of drug-resistant TB strains poses a severe public health threat, necessitating the development of new and effective therapeutic agents. In recent years, the focus has increasingly shifted towards novel antibiotics with unique mechanisms of action that can overcome existing resistance patterns. Among these, the oxazolidinone class of antibiotics has garnered significant attention due to its potent anti-mycobacterial activity and promising clinical outcomes.
Oxazolidinones are a class of synthetic antibiotics that inhibit protein synthesis by binding to the bacterial 50S ribosomal subunit, thereby preventing the formation of a functional 70S initiation complex(7). This unique mechanism of action makes oxazolidinones effective against a wide range of Gram-positive bacteria, including drug-resistant strains. The first oxazolidinone approved for clinical use was linezolid, which has since been utilized in treating various infections caused by resistant bacteria, including TB(8-10). Linezolid's efficacy against MDR-TB and XDR-TB has been well-documented in clinical studies, showing the ability to improve treatment outcomes significantly. However, its use is often limited by adverse effects, particularly myelosuppression, peripheral neuropathy, and optic neuropathy, especially with prolonged use(11, 12). These side effects have prompted the development of newer oxazolidinones with improved safety profiles and enhanced anti-tubercular activity.
Recent advancements in the field have led to the discovery and development of several novel oxazolidinone compounds. These new agents are designed to retain the potent anti-mycobacterial properties of linezolid while minimizing its associated toxicities. For instance, in preclinical and clinical studies, tedizolid, a second-generation oxazolidinone, has demonstrated a more favorable safety profile and potent activity against Mtb(13, 14). Other promising candidates in various stages of development include sutezolid and delpazolid, which have also shown efficacy against drug-resistant TB strains with potentially fewer side effects. The clinical significance of novel oxazolidinone antibiotics extends beyond their direct antibacterial activity. These compounds are integral to combination therapy regimens that aim to shorten the duration of TB treatment, reduce the pill burden, and improve patient adherence. Effective combination regimens incorporating oxazolidinones can potentially transform the management of MDR-TB and XDR-TB, leading to higher cure rates and better overall outcomes.
In conclusion, TB remains a critical global health challenge, exacerbated by the rise of drug-resistant strains. The development of novel oxazolidinone antibiotics represents a significant advancement in the fight against TB, offering hope for more effective and safer treatment options. This review provides a comprehensive overview of the development, mode of action, toxicity, and anti-TB activity especially for drug-resistant TB of new oxazolidinones.
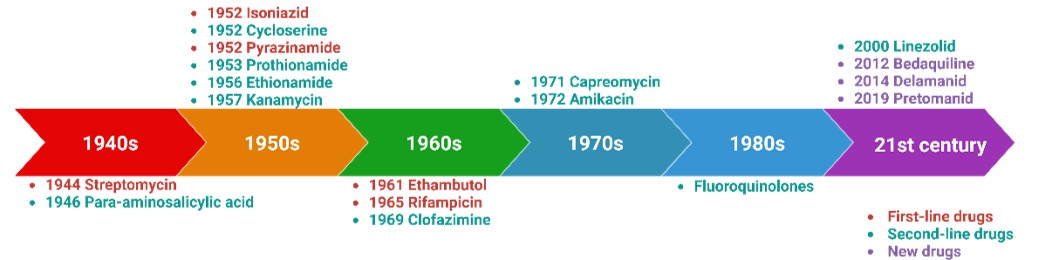
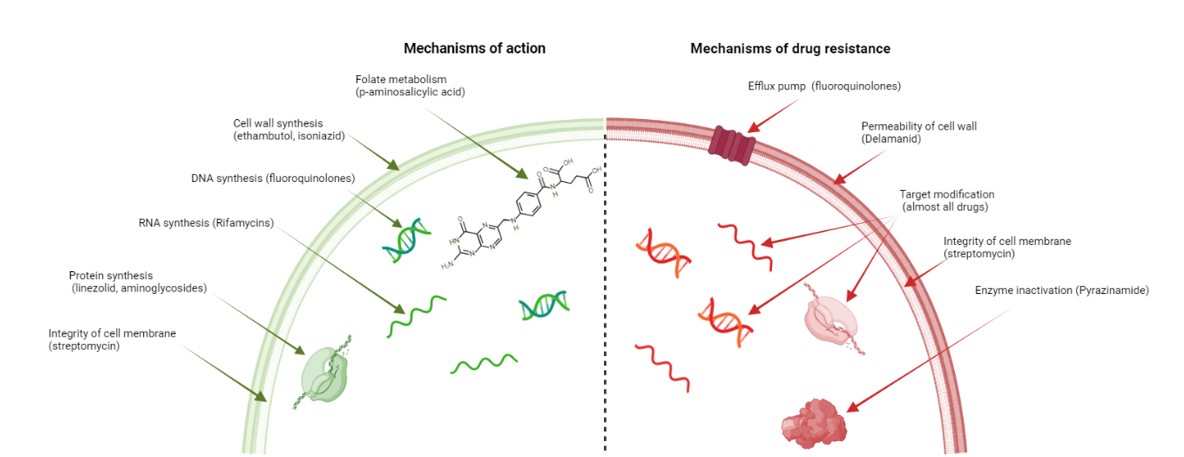
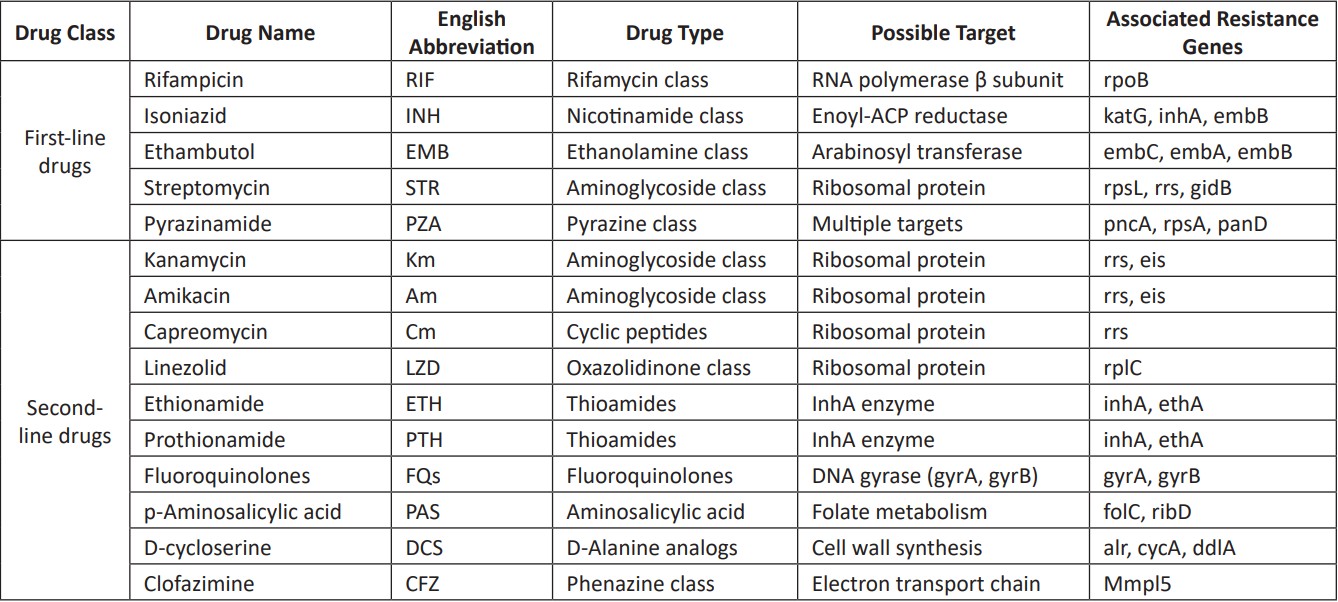
Isoniazid: Since its clinical application in 1952, isoniazid (INH) has always been a classic drug for tuberculosis treatment. It is one of the most common anti-tuberculosis drugs in clinical use because of its advantages of being cheap, effective, having few side effects, and bactericidal effects on both reproductive and static Mycobacterium tuberculosis(15). When INH enters MTB, it is first reduced by the catalase KatG. It binds to NAD+ to form the isonicotinic‐NAD complex (isonicotinic‐NAD), which inhibits the synthesis of mycolic acids in the cell wall of MTB by interacting with the NADH-specific enylacylapolipoprotein reductase InhA Integrity accelerates cell rupture and ultimately leads to the death of MTB in the metabolically active or dormant phase(16). In addition, INH has also been proven to be able to further prevent the occurrence of tuberculosis by reducing the death of immune cells induced by oxidative stress ROS(17). Studies targeting INH resistance have also focused on mutations in genes associated with InhA and KatG targets. For example,katG gene mutation, the most common of which is the mutation of the 315th Ser residue in KatG protein to Thr, can relieve the inhibitory effect of INH on the synthesis of myclonic acid and make MTB resistant to INH by promoting the synthesis of myclonic acid in the cell wall(18).
Similarly, the analysis of clinical INH-resistant strains found that there were a large number of inhA gene promoter mutations in drug-resistant strains, and the most common type of gene mutation was C15T, so it was speculated that inhA gene mutation may also cause MTB to develop resistance to INH(19). In addition, after INH enters the body, MTB may produce free radicals and affect its own metabolism, thereby reducing its sensitivity to INH. In addition, InhA protein Thr266 can be phosphorylated. After modification, its activity is reduced, and the permeability of the cell wall is changed, which may affect its resistance to INH(20). The acetylation modification of iron regulatory protein HupB also affects the expression of corresponding MTB genes, resulting in cell resistance to INH(21). In recent years, it has also been reported that the mutation of the embB gene related to ethambutol resistance may also cause MTB to INH mutation, and the specific mechanism needs to be further studied(22). In addition, PknG, SahH, KasB, and other proteins may also participate in generating INH resistance(23).
Rifampicin: Rifampicin (RIF) is a semi-synthetic antibiotic derived from rifamycin, which can inhibit both active and slow-metabolized MTB. RIF mainly acts on the subunit of RNA polymerase β of MTB, hinders mRNA extension, inhibits protein synthesis, affects transcription, and thus kills MTB(24). Long-term use of RIF injection in clinical treatment is prone to a certain degree of RIF resistance in patients. However, it was found in the actual situation that simple resistance to RIF was rare. In general, strains resistant to RIF are also resistant to other drugs (especially in combination with INH). Therefore, general RIF resistance is one of the signature characteristics of MDR. RIF mainly acts on the beta subunit of RNA polymerase, so RIF-induced drug resistance is usually associated with mutations in its coding gene rpoB(24). rpoB gene mutation causes conformation change of RNA polymerase subunit protein, resulting in RIF resistance. It is believed that the mutations associated with RIF resistance are mainly concentrated in the rifampicin resistance determining regions of the rpoB gene(25). According to WHO statistics, among the new cases in China in 2020, the percentage of TB cases confirmed by bacteriology to RIF is as high as 83%. Among the cases that recurred after treatment, the percentage of TB cases confirmed by bacteriology was as high as 97%(26). Therefore, it is often recommended to use RIF in combination with other drugs to prevent resistance and achieve better therapeutic effects.
Ethambutol: Ethambutol (EMB) was discovered in 1961 and used for the treatment of tuberculosis in 1966. EMB mainly acts on active growing MTB and has no obvious bactericidal effect on dormant MTB strains. The mechanism of action of EMB is related to the cell wall synthesis of MTB. As a structural analog of arabinose, EMB plays a bactericidal role by blocking the synthesis of arabinose galactose and reducing the permeability of the cell wall by inhibiting arabinose transferase during the cell wall synthesis of MTB (25). Studies have shown that the mechanism of EMB resistance is mainly related to mutations in arabinosyltransferase-related genes embC, embA, and embB. EmbB gene mutation was the most common in clinically isolated EMB-resistant strains. In addition, mutations in embC and EMBC-EMBA gene regions can also lead to drug resistance in EMB(27). Similarly, protein post-translational modification can indirectly regulate the transcription process of the emb ABC operon, thereby regulating the change of EMB resistance(27). Clinically, EMB is mainly used for the treatment of patients with contraindications to para-aminosalicylic acids and is often used in combination with INH and RIF.
Pyrazinamide: Pyrazinamide (PZA) is a structural analog of quinolinic acid, an important metabolite of NAD in Mycobacterium tuberculosis(28). PZA can kill dormant and semi-dormant MTB in host macrophages. It is believed that PZA enters bacteria via a membrane transport system and is converted to its active form, pyr‐azinoic acid (Pyrazinami-dase, PZase), by the pncA gene acid, POA). POA enters the extracellular acidic environment through the effection mechanism to form protonated pyrazinic acid, which causes cell damage by affecting the membrane potential and then kills MTB(28). In addition, PZA can also affect the normal metabolism of MTB by affecting dehydrogenase dehydrogenation, resulting in antibacterial effects(29). In 2011, Shi et al. found that the active form of PZA in vivo, POA, could directly target the S1 protein in the 30S subunit of the ribosome and affect the protein translation process, thereby enhancing the activity of PZA(29). At present, the mechanism of PZA resistance is still inconclusive, and studies mainly focus on pncA gene mutations(30, 31). In addition, rspA, panD, fadD2, and genes related to FAS‐ I metabolism may also be related to PZA resistance(30, 32).
Streptomycin: Streptomycin (STR) is the first injectable aminoglycoside drug used in the treatment of tuberculosis, which has a bactericidal effect on MTB in the logarithmic growth stage. STR can directly bind 16S rRNA(rrs gene coding) and S12 ribosomal protein (rspL gene coding) in the 30S subunit of MTB ribosome, hinder the binding of tRNA and ribosome, and then inhibit bacterial protein synthesis, resulting in bacterial death(33). Therefore, rspL and rrs gene mutations are one of the leading causes of STR resistance in MTB(34). It was found that 86.13% of STR-resistant strains carried rpsL or rrs gene mutation, and rpsL gene mutation accounted for the majority (67.15%). In addition, it has been reported that a low level of STR resistance is related to the mutation of the gidB gene encoding 16S rRNA methyltransferase(34, 35).
Fluoroquinolones: Fluoroquinolones (FQs) are widely used broad-spectrum antibiotics in clinical practice, and they are also the first choice drugs for the treatment of XDR tuberculosis. FQs commonly used in anti-tuberculosis treatment mainly include traditional ofloxacin, levofloxacin, moxifloxacin, and FQS. ciprofloxacin (ciprofloxacin) and the newer sparfloxacin (sparfloxacin). Clinically, FQs are primarily used for the combined treatment of drug-resistant tuberculosis due to the advantage that it is not easy to produce a cross-resistance reaction with other anti-tuberculosis drugs. For example, FQS has an excellent therapeutic effect on MDR-infected patients who are resistant to INH and RIF. FQs act on DNA rotase (type II DNASA topoisomerase) of MTB to inhibit its growth by inhibiting transcription and translation of MTB(36). Therefore, it is currently believed that FQs resistance sites are concentrated in the genes of quinolone resistance-determining regions (QRDRs)(36). Mutations in gyrA and gyrB genes encoding DNA rotase subunits lead to increased MIC of FQs. In addition, the increase of drug efflux pumps also affects the resistance of FQs; MTB cell wall thickening and permeability reduction are also the causes of FQs resistance, but the level of FQs resistance caused by cell wall thickening is low. The molecular mechanism needs to be further clarified(37).
P-aminosalicylic acid: P-aminosalicylic acid (PAS), a structural analog of the folic acid precursor p-aminobenzoic acid (PABA), is the most commonly used anti-tuberculosis drug in the folic acid antagonist class. Other folate antagonists include sulfoxone, WR99210, and cotrimoxazole (a combination of sulfamethoxazole and trimethopridine), targeting several enzymes necessary for the de novo synthesis of tetrahydrofolate: dihydrofolate synthetase synthase, DHFS, FolP1), Dihydro-folate reductase (DHFR), flavin-dependent thymidylate synthase, etc.(38). In MTB, PAS can competitively bind DHFS and inhibit the activity of DH‐FR, resulting in tetrahydrofolic acid, THF synthesis is significantly reduced, significantly inhibiting the conversion of deoxyuridine 5-phosphate (dUMP) to deoxythymidine 5-phosphate (dTMP), resulting in the disruption of DNA synthesis and death of MTB(39). At present, most studies on PAS resistance focus on the Folc protein coding gene folC, whose mutation will increase the metabolic rate of MTB to PAS, resulting in PAS resistance(40). Riboflavin biosynthesis enzyme (RibD) catalyzes dihydrofolic acid, DHF) and is transformed into THF. The mutation of its encoding gene ribD leads to the up-regulation of RibD expression to perform the corresponding function in place of DHFR, resulting in PAS resistance(41). In addition, mutations in protein-coding genes such as acyltransferase PapA1, SigB, and MmpL11 may also be related to the resistance of PAS, and the specific mechanism needs further study(42, 43).
Aminoglycoside antibiotics and peptide drugs: Aminoglycoside antibiotics and peptide drugs commonly used to treat tuberculosis include aminoglycoside kanamycin (Km), amikacin (Am), viomycin, and peptide capreomycin. These drugs inhibit the protein translation process by binding to 16S rRNA in the 30S subunit of MTB ribosomes, resulting in the death of MTB(44). Because of their similar mechanisms of action, the resistance mechanisms of these drugs are often discussed together. Not only that, but more and more studies are starting to look at cross-resistance between these drugs. Studies have shown that the tlyA gene may encode a predicted rRNA methyltransferase whose mutation enables MTB to develop resistance to capreomycin and violamycin(45). Mutations in the rrs gene encoding 16S rRNA were associated with resistance to all four drugs. Recent studies have shown that mutations in the promoter region of the eis gene encoding aminoglycoside acetyltransferase can affect the resistance of MTB to low levels of amikacin and kanamycin(46). In contrast, mutations in eis or whiB7 can lead to resistance of MTB to kanamycin(47). In clinical treatment, capreomycin has a good effect on TB at the rest stage. It has less toxic side effects than other second-line drugs. However, it is easy to produce drug resistance when used alone, so it is often combined with first-line drugs such as INH and EMB in clinical practice.
Linezolid: Linezolid (LZD) was approved by the FDA in the United States in April 2000 as the first fully synthetic oxazolidinone antibacterial drug, which can effectively treat various gram-positive bacterial infections. LZD acts on 23S rRNA of the 50S subunit of the ribosome and exerts anti-MTB effects by binding to the 50S subunit and inhibiting the formation of the 70S translation initiation complex, preventing protein translation(48). Currently, most genetic studies associated with LZD resistance have focused on rplC genes and rrl genes. The rplC gene encodes ribosomal protein L3 in the 50S subunit of the MTB ribosome. rplC mutations are often associated with varying degrees of LZD resistance, and the most common mutation is the 460th Thr mutation to Cys(49). The rrl gene mainly encodes 23S rRNA, and its mutation also causes MTB to develop LZD resistance. G2061T is the most common mutation, which will cause MTB strains to develop high resistance to LZD(50). In clinical treatment, LZD has strong side effects such as blood and myelotoxicity; about 40%-90% of patients suffer adverse reactions, and 6%-8% of patients stop taking LZD due to side effects such as intense neuropathy and myelosuppression(51). In the treatment of MTB, the more excellent toxic side effects make the clinical use of LZD strictly limited. However, as the first fully synthesized oxazolidinone antibacterial drug, its application in treating tuberculosis provides a way to explore new oxazolidinone anti-MTB drugs. In a clinical randomized trial by Conradie et al., the efficacy and safety of different doses and durations of linezolid in a bedaquiline–pretomanid–linezolid regimen for drug-resistant tuberculosis were evaluated. They concluded that a 600 mg dose of linezolid for 26 weeks provided the best balance of efficacy (84% favorable outcomes) and safety, with fewer adverse events and dose modifications.
Ethioisonicotinamide: Ethioisonicotinamide (ETH) and propylthioisonicotinamide (PTH) are both thioamide drugs. They are similar to INH in action, but their antibacterial activity is much lower than INH, and they have partial cross-resistance with INH(52). Similar to INH, the action mechanism of ETH and PTH requires the activation of monooxygenase EthA protein to generate metabolites with a similar structure to isonicotinic acid, inhibit the activity of target protein InhA enzyme, destroy the synthesis of mycetonic acid in MTB cell wall, lead to the destruction of cell wall integrity, accelerate cell breakdown, and eventually lead to metabolic hyperactivity or quiescence MTB dies(16). Therefore, mutations associated with ETH and PTH resistance mainly change drug-activated ethA and drug-target inhA genes(53). In addition, transcription suppressor EthR inhibits the expression of ethA by binding to the ETHa-ETHR gene spacer region, reducing the activity of ethA protein and resulting in increased resistance of mycobacterium tuberculosis to ETH(54). Some studies have found that in the clinical treatment of MDR-TB patients, ETH and PTH resistance is more serious. Therefore, some scholars suggest that patients with a history of ETH and PTH should undergo drug sensitivity tests before using the two drugs again to avoid insufficient therapeutic effects caused by drug resistance(47).
D-cycloserine: D-cycloserine (DCS), like RIF, is a broad-spectrum antibiotic(55). DCS is a structural analog of D-alanine and mainly affects the synthesis of D-alanine peptidoglycan in the cell wall of MTB. By inhibiting the activities of alanine racemase (pncA gene coding) and alanine ligase (ddlA gene coding), it can damage cell wall synthesis and weaken the acid resistance of bacteria, thus playing a bactericidal or bacteriostatic role(56). The analysis of the known DCS resistance strains showed that the mutated genes that might be related to DCS resistance were mainly concentrated in ald, pncA, and ddlA genes. The gene ald encodes L-alanine dehydrogenase and is involved in alanine metabolism. The absence of ald leads to L-alanine compensation in MTB cells, and a large amount of L-alanine accumulation reduces the competitive inhibitory effect of DCS, resulting in drug resistance in MTB(57). The gene pncA encodes alanine racemase, and the gene ddlA encodes alanine ligase. The overexpression of alanine racemase and ligase caused by mutations of the two genes could lead to DCS resistance in BCG strains. The latest study found that among the 513 isolates of XDR,153 DCS-resistant strains had mutations in pykA, cycA, ald, and alr genes. Still, the specific mechanism needed further study(58).
Clofazimine: Clofazimine (CFZ), also known as chlorphenphenazine or Kefenamine, is an iminophenazine drug whose critical structural feature is the phenazine nucleus, which gives it highly lipophilic and efficient transmembrane permeability. Early CFZ was mainly used in treating leprosy; in recent years, it has been used in treating tuberculosis(59). The detailed mechanism of action of CFZ is still unclear. Still, it may inhibit the growth and reproduction of bacteria by inhibiting the killing effect of MTB-derived factors on phagocytes. It may also be related to intracellular oxidative stress, such as hydrogen peroxide, superoxide, or ROS(60). Some studies have found that mutations in the transcription regulator rv0678 gene of MTB can up-regulate the expression of efflorescent pump MmpL5, resulting in cross-resistance between CFZ and Bedaquinoline (BDQ)(61).
Oxazolidinones in preclinical/clinical trials as anti-Drug-Resistant TB agents
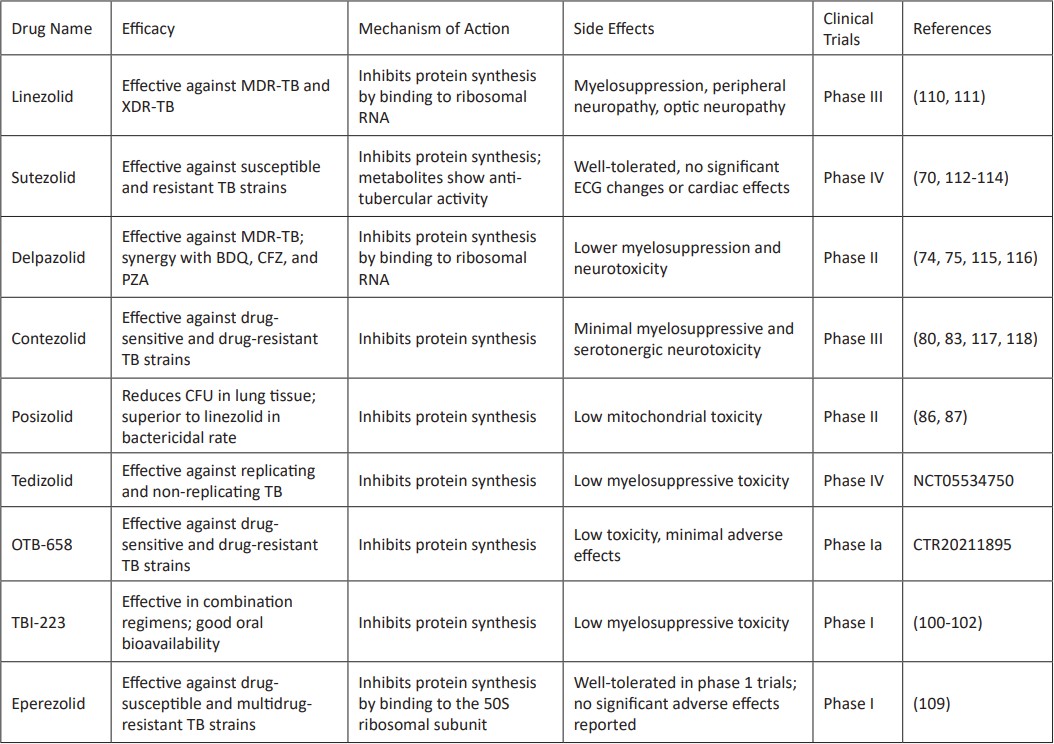
Sutezolid: Sutezolid, also known as PNU-100480, features a thiazole substituent in its chemical structure. It exhibits inhibitory activity against Mycobacterium tuberculosis in vitro, with a MIC range of 0.03 to 0.5 µg/mL for both susceptible and resistant strains. Its metabolites include the sulfoxide metabolite PNU-101603 and the phenyl sulfone metabolite PNU-101244, with PNU-101603 also displaying anti-tubercular activity(67). In vivo studies indicate that, after four weeks of administering Sutezolid at doses of 25 to 100 mg/kg, there is a significant reduction in infected mice's Mycobacterium tuberculosis colonies in the lungs and spleen, with efficacy comparable to INH(68). Mouse studies have shown that Sutezolid at 100 mg/kg has stronger anti-tubercular effects and better safety than linezolid. Additionally, Sutezolid enhances the anti-tubercular activity of the INH+RFP+PZA and MFX+PZA combinations, reducing bacterial load in mouse lungs by approximately 2log10CFU(69). Pharmacokinetically, Sutezolid is characterized by rapid oral absorption, with Tmax between 1.75 and 2.5 hours. It distributes quickly throughout the body, with a volume of distribution (VZ/F) ranging from 990 to 2000 L, a clearance rate (CL/F) of 145 to 167 L/h, and a half-life (T1/2) of 4.08 to 11.7 hours. The volume of distribution and half-life increase with higher doses, highlighting the central compartment's role in drug elimination. Sutezolid has a high safety profile; healthy subjects tolerated doses up to 1000 mg/kg(70). In single doses of 300 to 1800 mg, no significant ECG changes or other cardiac-related adverse effects were observed in healthy volunteers(71). Currently, a Phase IIb clinical trial is evaluating the safety, tolerability, pharmacokinetics, and dose-response relationships of various Sutezolid doses (0, 600 mg once daily, 1200 mg once daily, 600 mg twice daily, 800 mg twice daily) in combination with BDQ+DEL+MFX(72), aiming to determine the optimal Sutezolid dosage by examining the relationship between drug exposure and toxicity, ensuring maximal efficacy while maintaining acceptable safety levels.
Delpazolid: Delpazolid, also known as LCB01-0371, was synthesized by Lego Chem BioSciences, Korea. The cyclic amiodarone structure is applied to the critical skeleton of Delpazolid, which retains some hydrophobicity and has a weak alkaline pH similar to that of carboxylic acids. Therefore, In human physiological conditions, it can be charged by protons from the carboxylic acid to enhance its solubility and pharmacokinetic characteristics(73). Delpazolid MIC of MDR-TB, lower than the LZD with anti-tb activities within macrophages(49, 73), drug interaction research results show that Delpazolid and BDQ CFZ and PZA exist some synergy, and at the additive effect, fluoroquinolone drugs exist(73), in 400-1200 mg dose range, AUC0-12 and Cmax according to proportional rise, dose not show the drug characterized by nonlinear characteristics of PK(74), The oral absorption of Delpazolid is fast, the half-life of T1/2 is about 2h, which is only 1/3 to 1/2 of the half-life of LZD. The urinary excretion of delpazolid accounts for about 8% of the administered dose, and delpazolid can not be detected in the human body after 12h of administration(75). Delpazolid has a higher safety profile, showing lower myelosuppression and neurotoxicity in the rat model. It has demonstrated low mitochondrial toxicity in both phase Ⅰ clinical trials and preclinical studies, and because delpazolid is not a substrate for cytochrome P450 and drug transporters, it causes few drug-drug interactions(73, 76). Clinical trials (phase Ⅱ clinical trials) evaluation under the different doses of Delpazolid (800 mg once daily, 400 mg twice a day, 800 mg twice a day and 1200 mg once daily) early bactericidal activity and safety. The average daily colony counts were 0.044±0.016,0.053±0.017,0.043±0.016 and 0.019±0.017, respectively. No serious adverse reactions related to the drug were found in the subjects(75). Delpazolid is undergoing a phase 1b clinical trial to evaluate the relationship between the drug effect in the combination of BDQ+DEL+MFX and the drug exposure in vivo(75).
Contezolid: Contezolid (MRX-1) on the structure of the "three fluorine non coplanar" molecular structure design, namely the B ring in F, added A, B ring non coplanar, significantly reduce toxicity; C ring two alternative morpholine ring hydrogen pyridine ketone structure, speed up the drug metabolism and short half-life (77), microporous plate dilution in vitro experiments found contezolid sensitive to isoniazid and drug-resistant mycobacterium tuberculosis strains have inhibition, MIC between 0.5-1.0 μg/m L, its activity is similar to LZD(78), in mycobacterium tuberculosis Erdman infected BALB/c mice model, 100 mg/kg dose conetezolid and LZD can significantly reduce the lung tissue in mice, showed a greater resistance to tuberculosis activity, low dose (25 mg/kg, the BID) and medium doses (50 mg/kg, BID) contezolid bacteriostasis of high dose group of weaker(78), population pharmacokinetic studies have shown that oral ADME contezolid process is the second chamber model, absorption process influenced by food, the subjects weight affect peripheral distribution volume, Contezolid pseudo-first-order eliminate dynamics characteristics, mainly through urine and faeces, and the primary metabolites of MRX445-1 and MRX459 (79, 80), contezolid security is good, can tolerate oral single-dose 800 healthy subjects, 1200 and 1600 mg(81), PK/PD studies showed that contezolid did not prolong the QT interval at the therapeutic dose of 800 mg, while at the off-therapeutic dose (1600 mg) had only mild effect on QT interval prolongation (82, 83), in addition, it also showed weak induction of MAO-related serotonergic neurotoxicity and myelosuppressive toxicity(77).
Posizolid: Posizolid (AZD5847) anti-tb activity was dose-dependent, both inside and outside the cells with anti-tb activities, MIC < 1 μg/mL. AZD5847 was superior to LZD regarding anti-tuberculosis activity and bactericidal rate(84). Four weeks after treatment with AZD5847, the tuberculosis-infected model mice could reduce 1log10 CFU in lung tissue (AUC between 105 and 158 μg/mL). PK/PD studies have shown that AZD5847 pharmacodynamics target value in AUC/MIC > 20 free drugs, in the effective drug combination scheme % T > MIC25%(85), AZD5847 and other anti-tuberculosis drugs applications have additive effect, prompt AZD5748 can be used in the combination therapy in the future. Clinical trial at a later stage 1 in the study, evaluation AZD5847 is sensitive to infection of 60 strains of TB patients in the early bactericidal activity of 14 d, results show that the AZD5847 has shown superior to other anti-TB activities of Oxazolidinone compounds. From the pharmacokinetic characteristics analysis, AZD5847 distribution in the human body according to the second chamber model, the existence of the phenomenon of drug absorption process lag, and AZD5847 distribution volume in mice and the clearance is low, and elimination half-life is longer than the other. AZD5847 AUC, MIC (fAuc), and f T> MIC three PK/PD parameters are lower than the positive control drug LZD and sutezolid(86, 87). Astra Zeneca company currently has compound AZD5847 removed from the new drug pipeline.
Tedizolid: As an oxazolidinone prodrug, tedizolid phosphate can be rapidly converted into tedizolid with antibacterial activity in vivo(88). The C-5 side chain of tedizolid was modified, and the D loop was added to increase the binding site with the ribosome, thereby increasing the antibacterial activity(89). In vitro experiments showed that tedizolid had both sensitive and resistant clinical strains of Mycobacterium tuberculosis. It also has an inhibitory effect on Mycobacterium tuberculosis in the non-replication phase and has an intracellular activity, which is comparable to the positive control drugs MFX and RFP(90-92). Tedizolid can form an effective drug combination with existing anti-tuberculosis drugs. Studies have shown that the drug combination of tedizolid instead of LZD and MFX has a constant bactericidal rate (0.27±10.05) daily(93, 94), tedizolid has a stronger affinity for targets, whose resistance than LZD is less(95), tedizolid has high safety, in vitro experiments show as tedizolid higher doses, no increase of THP-1 cells toxicity. Tedizolid at AUC0-24 ≤ 90 mg·h/L cases did not cut mitochondrial enzyme gene expression, prompting minor mitochondrial toxicity (93). For TB patients with liver transplantation, tedizolid did not cause anemia and gastrointestinal adverse reactions after 20 months of continuous use, indicating the high safety of tedizolid in the case of long-term use(96). Phase Ⅱ clinical trial (NC clinical trials T05534750) aimed to evaluate the early bactericidal activity of tedizolid in treatment-sensitive patients with pulmonary tuberculosis, which is currently in the recruitment phase.
OTB-658: OTB-658 is optimized on the chemical structure of LZD by introducing the mercaptolin substituent to increase its chemical structure's steric hindrance and enhance the compound's metabolic stability in vivo (97). OTB-658 MIC is lower than the positive control drug LZD; for standard mycobacterium tuberculosis strain H37Rv, clinically isolated sensitive and resistant strains, it also has good activity. OTB-658 has intracellular activity and can enter macrophages to inhibit the growth of M. tuberculosis. In addition, the OTB - 658 spontaneous mutation frequency is low, about 10-8; prompt OTB - 658(97, 98) is not easy to result in a drug resistance situation. The in vivo metabolites of OTB-658, OTB-665, and OTB-698 also have anti-tuberculosis activity, inhibiting the growth of M. tuberculosis (MIC, 0.44 and 0.93μg/mL, respectively)(99). In acute and chronic tuberculosis infection in mice models, low doses of OTB-658 showed stronger anti-TB activity than medium-dose linezolid. OTB-658 low dose group (25 mg/kg) in mice lung, spleen tissues colony count (colony forming units, CFU) were lower than LZD group (50 mg/kg), and the difference was statistically significant(97, 98). Preclinical studies have shown that compound OTB-658 has good pharmacokinetic characteristics, such as rapid oral absorption, high bioavailability, and long half-life. The cytotoxicity was low, which made it difficult to cause adverse reactions of bone marrow suppression; peripheral neurotoxicity and cardiac toxicity were low, and no adverse reactions were found to significantly reduce white blood cells, platelets, and other blood cells. In addition, OTB-658 has good druggability and high stability in microsomes, no inhibition on CYP 450 was observed in experiments, and drug-drug interaction is not easy to occur, which is suitable for future clinical application to form an effective combination drug regimen with other drugs(97). In January and February 2011, a Phase I clinical trial of OTB-658 (registration number CTR20211895) was completed, evaluating the tolerability and pharmacokinetics of OTB-658 in a single administration in healthy subjects.
TBI - 223: The antibacterial activity of TBI-223 is comparable to that of linezolid. In the experiment to evaluate the drug combination regimen in the mouse tuberculosis infection model, when Linezolid in the BPaL combination regimen was replaced with TBI-223, the combination regimen still showed good anti-tuberculosis activity and ability to inhibit recurrence, and its antibacterial activity was positively correlated with its dose(100). Regarding pharmacokinetics, TBI-223 has a high oral bioavailability in rats and dogs. At the same time, TBI-223 has high stability in liver microsomes and has no inhibition or induction effect on five cytochrome enzymes and no activation effect on nuclear receptors PXR, CAR, and AhR that can mediate cytochrome enzyme activation(101). Of concern is that TBI-223 has a high safety profile and low myelopathic toxicity, and no hematological changes or myelopathic toxicity were observed in rats in a 28-day rat toxicity study. Its low inhibitory ability on mammalian mitochondrial protein synthesis suggests that TBI-223 does not interfere with human mitochondrial protein synthesis during its anti-tuberculosis activity in vivo and is not prone to adverse reactions similar to LZD's inhibition of human mitochondrial protein synthesis, which can improve the safety of clinical use(72, 100, 102). At present, TBI-223 has completed clinical trial phase I (NCT03758612).
Eperezolid: Eperezolid (U-100592) is an oxazolidinone antibiotic developed by Pharmacia & Upjohn Inc.(103). It represents an early attempt to address drug-resistant bacterial infections using the oxazolidinone class. It shares structural similarities with linezolid, a more widely known and utilized oxazolidinone. The primary structural difference between the two compounds lies in their side chains: eperezolid possesses a 4-(hydroxyacetyl) piperazine-1-yl group. At the same time, linezolid has a morpholin-1-yl group(104, 105). This subtle difference in structure can influence the pharmacokinetic and pharmacodynamic properties of the drug. Similar to other oxazolidinones, eperezolid inhibits bacterial protein synthesis by binding to the 50S ribosomal subunit. This binding action interferes with the assembly of the 70S initiation complex, which is essential for bacterial translation and protein synthesis. By disrupting this critical process, eperezolid effectively halts bacterial growth and proliferation(106, 107). Eperezolid has demonstrated potent in vitro activity against both drug-susceptible and multidrug-resistant Mycobacterium tuberculosis strains. MIC values for drug-susceptible strains range from 0.125 to 0.5 µg/mL, while MIC values for multidrug-resistant strains fall between 0.5 and 2 µg/mL. These findings indicate that eperezolid has the potential to be an effective treatment for various forms of tuberculosis, including those resistant to conventional therapies. In vivo studies, however, have been less encouraging. In murine models, eperezolid showed limited efficacy against M. tuberculosis. At a dose of 100 mg/kg, eperezolid only produced a minor reduction in TB organisms in the lungs and spleen after four weeks of treatment, compared to the more significant reductions observed with isoniazid and linezolid. This modest in vivo efficacy raised concerns about the drug's potential effectiveness in human clinical settings. Human pharmacokinetic studies revealed that eperezolid has a dose-proportional, linear increase in plasma steady-state area under the curve and maximum concentration values at single oral doses ranging from 50 to 1000 mg. Importantly, these studies indicated that eperezolid was well tolerated, with no significant adverse effects reported in the phase 1 trials. Despite its favorable safety profile, eperezolid's development was discontinued in favor of linezolid. This decision was based on linezolid's superior pharmacokinetic properties, including better oral bioavailability and tissue penetration, as well as its more pronounced efficacy in preclinical models(108, 109).
Summary and Prospect
Humanity has been fighting tuberculosis for centuries, and the drugs to treat tuberculosis have made good progress at this stage. However, there are still many problems that need to be solved. From the current research, our understanding of the mechanism of MTB resistance is still limited, which adds difficulty to developing new clinical anti-TB drugs. The launch of new drugs will undoubtedly bring more opportunities for TB patients to cure. However, the price of new drugs is still costly. According to the latest report statistics of WHO, among the patients successfully cured, 94% have been treated for up to 2 years, and 4% have been treated for up to 3 years. The long treatment cycle leads to poor medication compliance and further aggravates the emergence of TB drug resistance. Therefore, developing new drugs and new formulation types to shorten the treatment time is also urgent. Understanding the mechanism of MTB resistance is crucial for the development of new anti-TB drugs. The rapid development of molecular diagnosis and omics analysis has also provided a lot of help for the identification of drug resistance genes. However, there are still many problems, such as whether there is interaction between different drug resistance mechanisms, whether cross-resistance leads to the accumulation of drug resistance mutations or antagonism, and whether competition between drugs under the same drug resistance mechanism is still worthy of research topics. In this review, the antimicrobial mechanism of anti-tuberculosis drugs and the drug resistance mechanism of Mycobacterium tuberculosis are described to provide a reference for developing more new drugs.
In summary, oxazolidinone candidates have many unique advantages, including unique targets, no cross-resistance with existing anti-tuberculosis drugs, and can reduce the emergence of drug-resistant strains. After structural modification, the drug has a stronger affinity with the target, better pharmacokinetic characteristics, rapid oral absorption, and high tissue permeability, and can be rapidly and thoroughly degraded in vivo without accumulation. The oral bioavailability of the drug is higher, which can better exert anti-tuberculosis activity in vivo and enhance the antibacterial activity of the drug combination regimen. At the same time, it has improved safety, low mitochondrial toxicity, low peripheral neurotoxicity, and myelosuppressive toxicity, and it does not interact with drug metabolism enzymes, reducing the occurrence of adverse reactions. These advantages give oxazolidinone drugs broad development prospects and potential in the field of anti-tuberculosis drugs. Although oxazolidinones have made great progress in the research of anti-tuberculosis drugs, there are still some challenges and problems. At present, most of the oxazolidinones are in multiple clinical trials to evaluate the efficacy and safety of the treatment of skin and soft tissue infections, sepsis, bone marrow inflammation, and other diseases caused by gram-positive bacteria, while in the treatment of drug-resistant tuberculosis, most of the oxazolidinones are still in the laboratory research and early clinical trials, mainly in healthy subjects to evaluate safety and tolerability The dosing of oxazolidinones is explored in fixed drug combinations. At present, few clinical trials have been conducted on the early bactericidal activity and therapeutic efficacy of drugs. Future studies on the toxicity, pharmacokinetics, mechanism of action, and drug resistance of oxazolidinones need to be further studied, and clinical trials of oxazolidinones need to be accelerated.
Author Contributions: QZ wrote the original and revised manuscripts and created the figures and tables. TL provided assistance in writing and conceptualization and also helped to develop the scope and depth of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding: This study received funding from the Sichuan Science and Technology Agency(2022NSFSC0845) and the National Natural Science Foundation of China(No. 82302690)
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Data Availability Statement: No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments: We would like to take this opportunity to express our sincere gratitude to the West China Hospital, Sichuan University for their strong support of this research. Figure 1 and Figure 2 were created using BioRender.com (accessed on 20 Jul 2024).
Conflicts of Interest: The authors declare no conflicts of interest.