Sensitivity of Bud Rot (Phytophthora sp.) Isolated from Coconut Palms in Jamaica to local Commercial Fungicide Pilarzox and Turmeric powder.
Curtis Hanchard*; Wayne Myrie
Research Department, Coconut Industry Board, Kingston, Jamaica, West Indies
*Corresponding author
Curtis Hanchard, Coconut Industry Board, 18 Waterloo Road, Kingston 10, Jamaica, West Indies
DOI: 10.55920/JCRMHS.2025.11.001468
Table 2: Fungicide inhibition of phytophthora using Pilarzox at various Cymoxanil concentrations
Table 3: Overall fungicide inhibition of Phytophthora using Pilarzox
Table 4: Fungicide Inhibition of Phytophthora Using Turmeric at Various Concentrations
Cyazofamid is generally applied at 80-100 g active ingredient/Ha or 50-100 ppm concentration. In vitro testing using Pilarzox showed a 38.9% inhibition at 100 ppm active ingredient on Phytophthora sp. There was a gradual increase in inhibition by Cyazofamid with complete inhibition observed at 500 ppm after which there is a plateau. Therefore, complete inhibition is achieved at approximately 500 ppm of the active ingredient Cyazofamid. The difference between the result observed (~500 ppm for complete inhibition) and the result using the recommended concentration of Cyazofamid (50-100 ppm), may be due to Phytophthora developing resistance to Cyazofamid. Ivanov et al, 2021 observed that continuous mass application of fungicides causes increased evolutionary pressure on P. infestans and consequently initiates rapid adaptation and acquisition of resistance to the fungicide involved. Cyazofamid is present in a few commercial fungicides, therefore the resistance may be present in the local population. Thus, a higher concentration of Cyazofamid is required to achieve the desired effect. Focusing on Cymoxanil, it was observed that a very high concentration of 1000 – 5,000 ppm was required to achieve complete inhibition of Phytophthora. Pilarzox optimum average inhibition was observed to be 1000-5,000 ppm. Observed results suggest the possible development of resistance of the local Phytophthora sp. to Cyazofamid and Cymoxanil-based fungicides in the field.
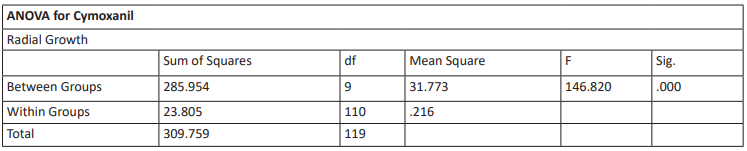
Cymoxanil is commonly used in the treatment of phytophthora such as P. Infestans. Despite its widespread use in agriculture, the exact mechanism of action remains unclear (Kazmirchuk et al, 2024). A study by Kazmirchuk et al, 2024 suggests that the Cymoxanil mode of action is the inhibition of dihydrofolate reductase enzyme activity in a dose-dependent relationship. It was observed that for complete inhibition focusing on the active ingredient Cymoxanil, high inhibition was observed at 200 ppm of 77%. This increases up to 81% at 500 ppm. Complete inhibition is only observed at 1000-5,000 ppm (see Table 2). Therefore, Phytophthora may also be experiencing increased resistance to Cymoxanil as well.
Some Phytophthora isolates exhibit increased expression of efflux transporters or enhanced metabolic detoxification. These mechanisms help remove or neutralize the fungicide, allowing the pathogen to survive even in the presence of chemical treatments (Pasteris et al., 2000). The resistance observed may also be associated with mutations in genes encoding the target enzymes of fungicides. For instance, alterations in the amino acid sequence of key enzymes can reduce the binding affinity of phenylamide fungicides, thereby diminishing their efficacy. As well as changes in the metabolic pathways of Phytophthora may also contribute to fungicide resistance. Such alterations can enable the pathogen to bypass the inhibitory effects of fungicides, ensuring continued growth and reproduction (Gisi & Sierotzki, 2008).
Curcuma longa, commonly called Turmeric is a tuberous rhizome known for its antioxidant and antimicrobial properties of its naturally occurring phenolic compounds (Murugesh et al, 2019). It’s believed that turmeric extract inhibits fungal growth by alteration in the morphology of the hyphae which may appear severely collapsed, plasma membrane disruption, mitochondrial destruction, lack of cytoplasm, folding of the nuclear membrane and thickened cell wall caused by chemical components of the spice extract (Murugesh et al, 2019). Martins et al 2009 study showed that Turmeric could inhibit the growth of a wide variety of fungal species. While Chen et al. (2018) identified several active compounds that contribute to its antifungal activity through multiple mechanisms. The research identified Curdione as one of turmeric's most potent antifungal agents. It has demonstrated strong activity against pathogens such as Fusarium graminearum, a fungus responsible for significant crop diseases. Curcumin, the most well-known compound in turmeric, demonstrated higher efficacy than conventional antifungal agents like fluconazole against pathogens such as Paracoccidioides brasiliensis, which causes paracoccidioidomycosis. Curcumin can damage the fungal cell membrane, impairing its function and leading to cell death. β-elemene contributes to the overall antifungal effectiveness of turmeric extracts, reinforcing the effects of curdione and curcumin. Turmeric essential oil contains several volatile compounds, including eucalyptol, β-pinene, and camphor, which have been found to inhibit the growth of fungi such as Aspergillus flavus, a common contaminant in food products. Turmeric’s antifungal activity operates through multiple mechanisms, one of which is the disruption of mitochondrial function. Compounds in turmeric can interfere with ATP generation in fungal cells by targeting key mitochondrial enzymes such as succinate dehydrogenase (SDH) and NADH oxidase. This energy depletion inhibits fungal growth (Oza et al., 2021).
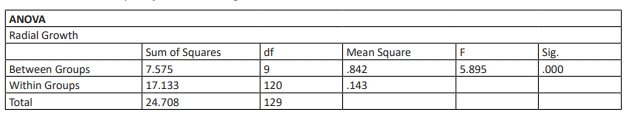
After the assessment of Turmeric on isolated phytophthora sp., it was proven that there was a significant difference between samples and control at 100, 200, 5,000 and 10,000 ppm (see table 19,20,21,22). The greatest inhibition observed was 12% at 10,000 ppm of Turmeric extract (see table 4). This may be because dry turmeric powder was used which may have affected the curcuminoids in the extract. Turmeric as a stand-alone fungicide shows low effectiveness against Phytophthora. However, as an organic fungicidal cocktail, it may improve its effectiveness. Alternatively, fresh Turmeric may have significant findings compared to the Turmeric powder used. Further research is needed for Turmeric as an organic fungicide as it shows promise as a growth inhibitor of Phytophthora.
Table 6: Multiple Comparison Analysis of Cyazofamid using SPSS
Table 7: ANOVA Analysis of Cyazofamid using SPSS
Table 8: Tukey HSD Analysis of Cyazofamid using SPSS
S2 Appendix
Table 9: Analysis of Cymoxanil using SPSS
Table 10: ANOVA Analysis of Cymoxanil using SPSS
Table 11: Multiple Comparison Analysis of Cymoxanil using SPSS
Table 12: Tukey HSD Analysis of Cymoxanil using SPSS
S3 Appendix
Table 14: Analysis of Pilarzox using SPSS
Table 15: ANOVA Analysis of Pilarzox using SPSS
Table 16: Multiple Comparison Analysis of Pilarzox using SPSS
Table 17: Tukey HSD Analysis of Pilarzox using SPSS
S4 Appendix
Turmeric results
Table 19: Analysis of Turmeric using SPSS
Table 20: ANOVA Analysis of Turmeric using SPSS
Table 21: Multiple Comparison Analysis of Turmeric using SPSS
Table 22: Tukey HSD Analysis of Turmeric using SPSS