The Tip of the Iceberg: Cerebellar Stroke in a Young Patient with Dyspnoea
Bazo Medina Jessica M*, Castaño Guerra Rodolfo de Jesus, Ayala Hernández Eduardo, Alvarado Ávila J. Cesar, Muñoz Reyes Pamela M
Hospital General de México “Dr. Eduardo Liceaga”, México City.
*Corresponding author
Bazo Medina Jessica M, Hospital General de México “Dr. Eduardo Liceaga”, México City
DOI: 10.55920/JCRMHS.2025.11.001467
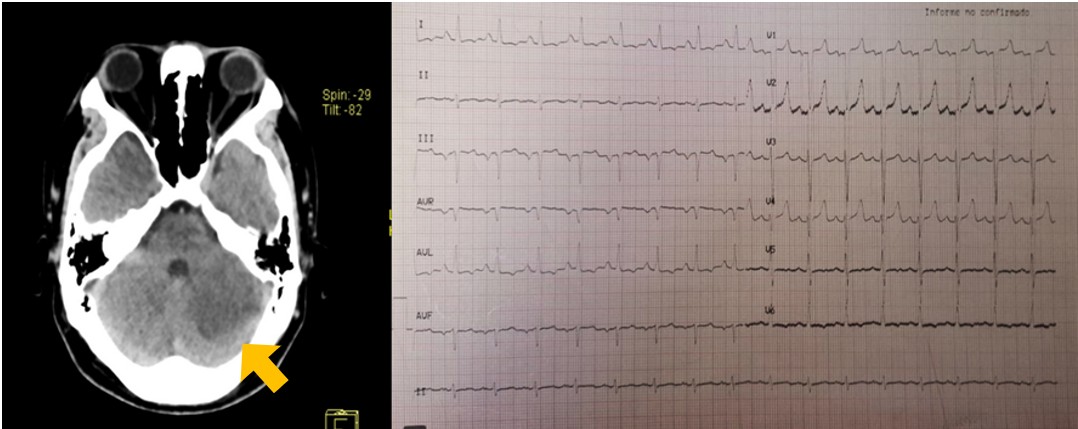
Figure 1: Left side: CT brain scan, an arrow shows the site of ictus at cerebellar hemisphere. Right side: 12-lead electrocardiogram of the patient, with sinus rhythm, axis deviated up to -30°, atrial enlargement and left ventricle hypertrophy, QRS measures 80ms.
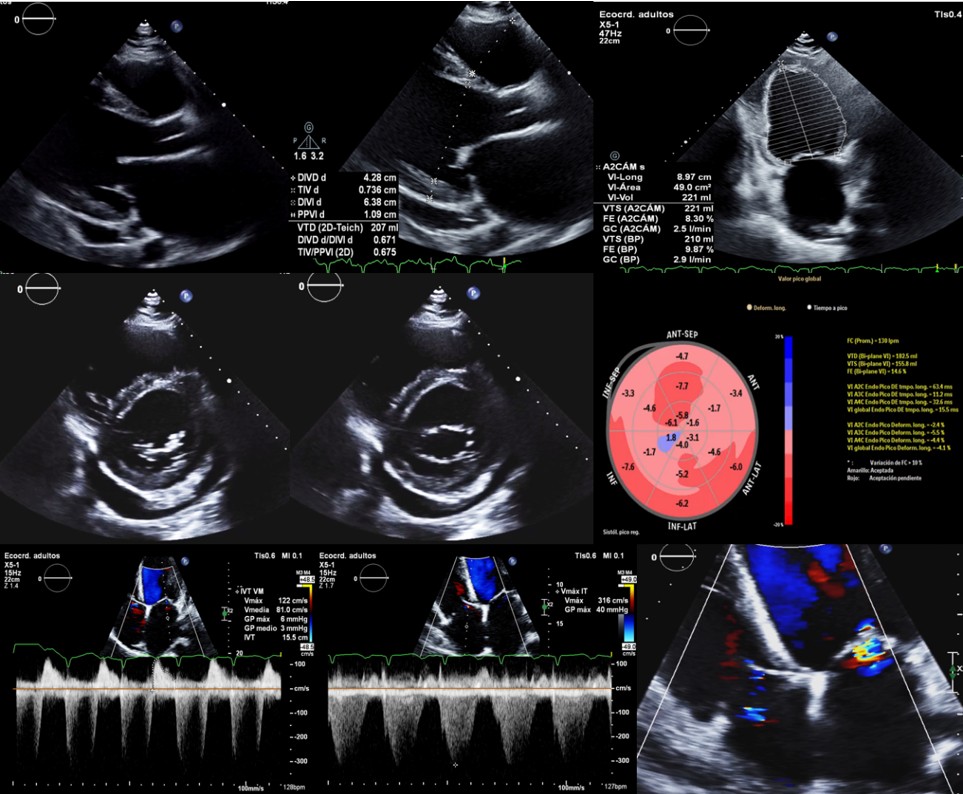
Figure 2: Transthoracic echocardiogram. Frozen images on PLAX, PSAX, and A2 showed significant enlargement of the left atrium, left ventricle, and right ventricle. The global longitudinal strain was -4.1% and the LVEF was 9%, with significant remodelling. Mitral and tricuspid regurgitation were observed in the context of dilated cardiomyopathy.
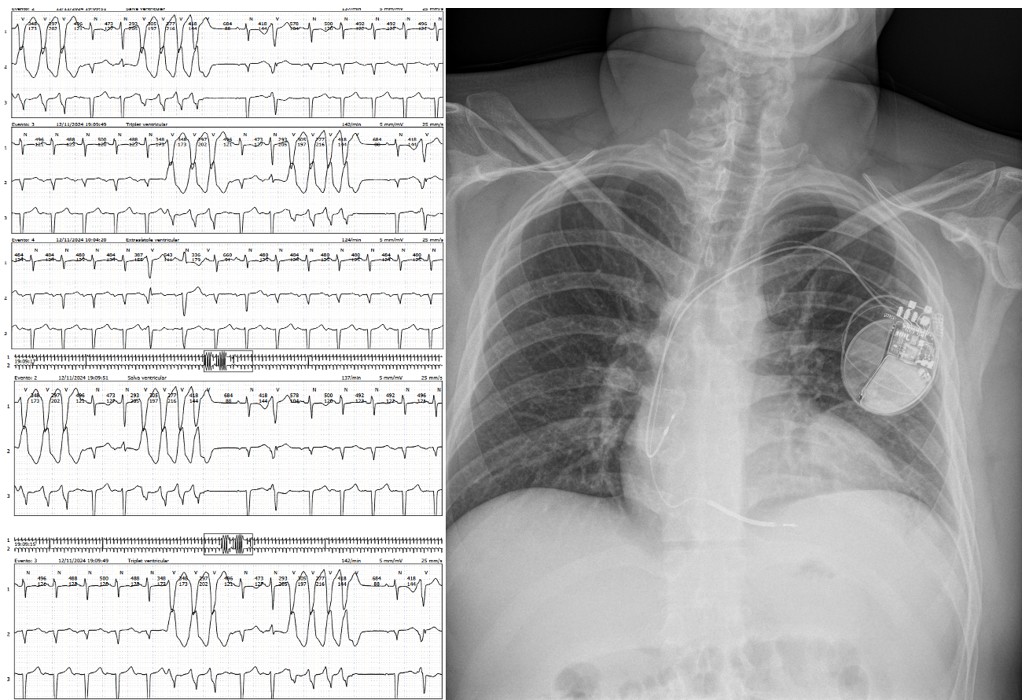
Figure 3: Non-sustained ventricular tachycardia was detected on Holter monitoring, and an implantable cardioverter-defibrillator was used to prevent sudden cardiac death.
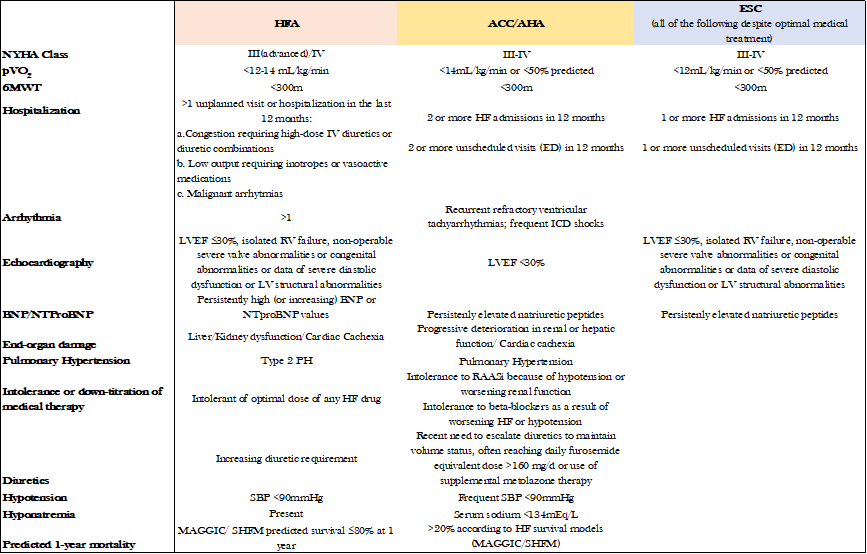
Table 1: Taken and modified from “Advanced heart failure: from definitions to therapeutic options”, by Garascia et al. (2023).
The physical examination most relevant findings were an irregular pulse, systemic venous congestion, an arrhythmic precordium with regurgitant mitral and tricuspid murmurs. However, the electrocardiogram showed sinus rhythm with evidence of left ventricular enlargement (Figure 1). A transthoracic echocardiogram was performed as a diagnostic approach, in which it was reported biventricular dilation, diastolic dysfunction, severely reduced systolic function (LVEF of 9% by biplane Simpson, GLS -4.1%, TAPSE of 12 mm, and CAF of 22%), in addition to a severely dilated left atrium, moderate functional mitral regurgitation, moderate tricuspid regurgitation, and mild aortic and pulmonary regurgitation (Figure 2). Also a Holter monitoring was ordered finding periods of sustained ventricular tachycardia.
Advanced heart failure, and in addition to statin treatment, he was started on guided medical therapy optimized to his tolerance. In the context of severely depressed EF and the arrhythmia identified during monitoring, an ICD was implanted (Figure 3). Also laboratory and imaging studies were performed in the etiological approach to dilated cardiomyopathy and as a heart transplant protocol.