How can Healthcare Providers Better Coordinate Care to Avoid Issues of Polypharmacy in Children with Autism Spectrum Disorder, considering both Non-Drug and Drug Treatment Strategies?
Dr. Khajik Sirob Yaqob Qazaryan*1, Dr. Noor Saadi Yousif Akash2, Dr Suwar Hassan Mohammed3 , Dr. Ali Alsarhan4, Dr Khaled Mohammed Al-mukdad5, Dr Taher Obaid Ali6
¹Specialist pediatrician, Department of pediatrics, child’s nutrition and growth, Zakho General Hospital. Specialist in child’s nutrition, growth with interest in pediatrics neurology, MBChB, MA, FRCPCH. Member of Kurdistan Pediatrics Society, Iraq. Full membership of ESPGHAN, Membership of American Dietetic Association, Member of Oxford University Hospitals
²Department of oral health and hygiene, clinical practitioner in dental health and surgery, B.D.S bachelor’s in dental surgery in Baghdad Al-Yarmouk University, Full member of Iraqis
³Clinical pharmacist, Board-certified in clinical pharmacy(KHCMS), Zakho Teachig Hospital, Kurdistan/Iraq.
⁴MD-Peds (SCFHS), CEBP, MRCPCH Pediatric Gastroenterology Fellow (F3)
⁵Specialist pediatrician, PhD, Damascus University, Bosra Alcham Hospital, MOH
⁶PhD, consultant Neurologist, Adult and Child Neurology, Mosul Teaching Hospital, MOH, Iraq.
*Corresponding author
Dr. Khajik Sirob Yaqob Qazaryan, Specialist pediatrician, Department of pediatrics, child’s nutrition and growth, Zakho General Hospital. Specialist in child’s nutrition, growth with interest in pediatrics neurology, MBChB, MA, FRCPCH. Member of Kurdistan Pediatrics Society, Iraq. Full membership of ESPGHAN, Membership of American Dietetic Association, Member of Oxford University Hospitals
DOI: 10.55920/JCRMHS.2025.10.001459
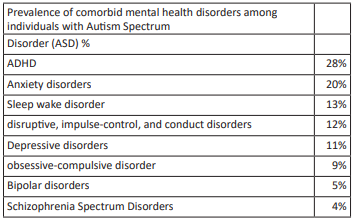
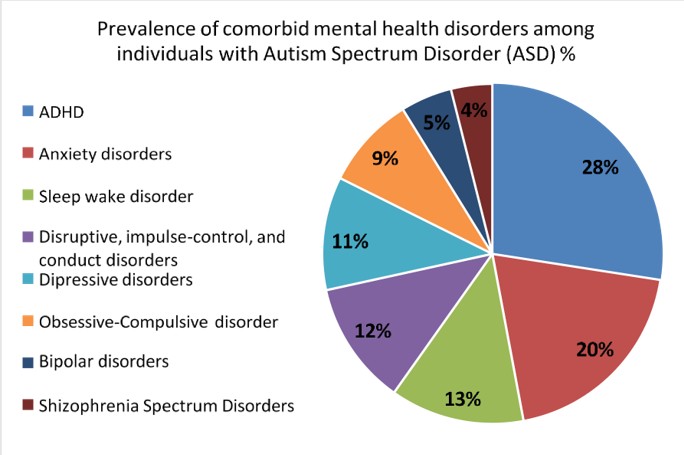
Figure1:
Non-drug treatment: Non-pharmacological interventions are recommended as first-line therapeutic approaches, especially for children aged seven or eight years and younger, aiming to support their developmental pathways. Various strategies are employed, including psychological therapies such as cognitive behavioral therapy, narrative therapy, schema therapy, and positive behavior support(3). Additionally, occupational therapy plays a vital role in enhancing fine and gross motor skills, interceptive awareness, and social skills, thereby contributing to the overall developmental progress of these children(8).
Drug-treatment strategies: Pharmacological interventions are crucial adjuncts to non-pharmacological therapies and are vital in effectively managing the myriad comorbidities frequently associated with ASD. According to (9), historically, traditional antipsychotics, most notably haloperidol, have formed the foundation of pharmacotherapeutic strategies for individuals with ASD. However, as diagnostic definitions have evolved and our understanding of the disorder deepened, atypical antipsychotics have emerged as superior alternatives, demonstrating enhanced efficacy and more favorable side effect profiles in pediatric populations with co-occurring psychiatric conditions(10).
A comprehensive review of the literature reveals a significant body of evidence comprising numerous small- scale studies, case reports, and open-label trials that underscore the efficacy of various pharmacological agents in the management of children diagnosed with ASD(11).
In the initiation of pharmacotherapy, it is imperative to adopt a methodical and systematic approach. Commencing treatment with a low dose is essential to allow for precise monitoring of patient responses while simultaneously minimizing the potential for adverse effects. The Centre for Interventional Pediatrics Psychopharmacology and Rare Diseases in the United Kingdom advocates for initiating treatment at doses ranging from one-eighth to one-sixth of standard adult levels. Such a strategy ensures that practitioners can make informed adjustments based on clinical response and tolerability after five to six half-lives of the medication. In the context of Australian practice, dose titration typically occurs every three to seven days; however, fluoxetine necessitates a more prolonged titration period due to its active metabolite, norfluoxetine, which has a half-life of approximately 9 to 14 days.
Clinicians must be acutely aware of the significant risk of polypharmacy among patients with ASD,(12) a concern evidenced by findings indicating that 34% of individuals receiving pharmacological treatment are subjected to multiple medications simultaneously(13).
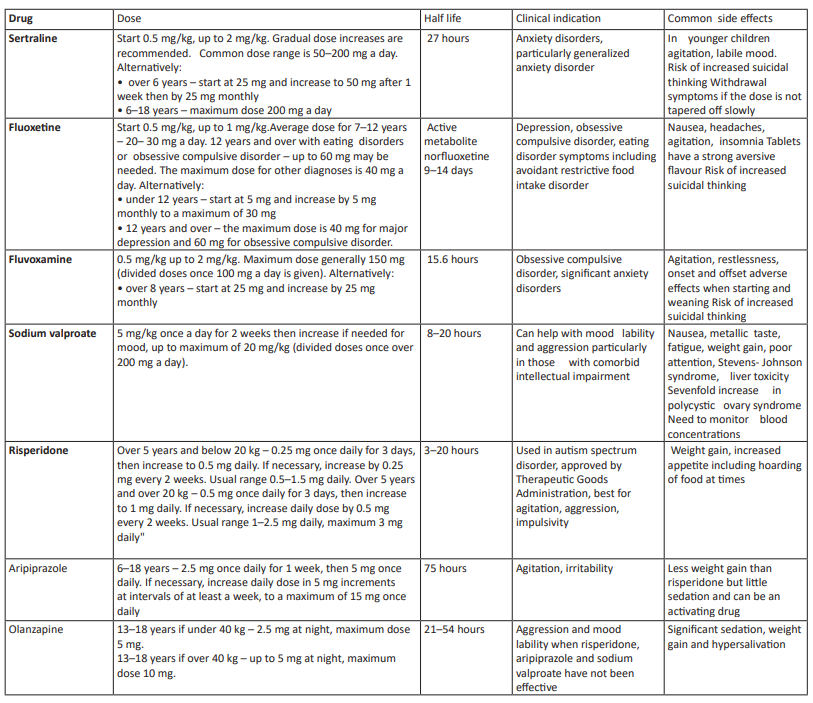
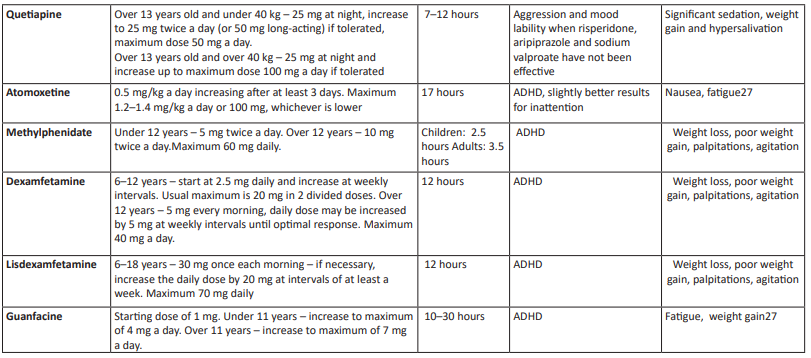
This heightened risk underscores the complexity of ASD, which often presents with a diverse spectrum of symptoms. Consequently, achieving comprehensive symptom relief with a single pharmacological agent is frequently unfeasible. The accompanying table provides a detailed overview of various pharmacological options tailored for specific clinical manifestations of the disorder, thereby facilitating a nuanced and targeted treatment strategy(14-17).
In addition, Gingival hypertrophy has long been recognised as an adverse effect of chronic phenytoin therapy [Gerret and Gerret, 2014; Ercoli, 2015; Thomason et al., 2020]. For other AEDs, such as valproate, oxcarbazepine, levetiracetam, topiramate, lamotrigine, this aspect has not been investigated sufficiently or the old studies claim no oral adverse effects (30,31,32).
It is crucial that the prescription of antipsychotic medications be carried out exclusively by qualified psychiatrists or pediatricians, or under their direct consultation, to ensure therapeutic effectiveness and safeguard patient safety. Ongoing and rigorous monitoring is absolutely essential, requiring comprehensive evaluations of weight, fasting lipid profiles, blood glucose levels, prolactin concentrations, and liver function. These assessments should be conducted biannually, with particular attention paid to the initial treatment phases, specifically at one month and three months post-initiation. This structured, evidence-based approach is not merely recommended; it is vital to optimizing clinical outcomes while ensuring the development of a comprehensive, responsible treatment paradigm for children diagnosed with ASD.
Ultimately, these practices will significantly enhance the overall well-being and quality of life for this vulnerable population.
Anxiety and depression: Anxiety is frequently identified as a prevalent comorbidity within the (ASD) population, often linked to significant impairments in social communication. Such impairments may induce considerable internal distress, particularly in contexts involving group interactions, exposure to novel environments, and adjustments to changes in routine. Empirical evidence supports the efficacy of talk-based interventions, especially cognitive- behavioral therapy (CBT), in the treatment of anxiety symptoms among individuals with ASD(18). However, the implementation of these therapeutic approaches may be complicated in cases of severe ASD, where individuals often experience profound difficulties forming therapeutic alliances and may find the articulation of distressing emotions to be intolerable.
In scenarios where in anxiety substantially disrupts the individual's functional capacities—such as through school avoidance, the deterioration of interpersonal relationships, or the cessation of previously engaging activities—pharmacological interventions can be warranted. Selective serotonin reuptake inhibitors (SSRIs), including sertraline, fluoxetine, and fluvoxamine, are generally regarded as first-line pharmacologic options for the treatment of anxiety in this cohort(19). Although fluvoxamine is acknowledged as a potential treatment, its profile of adverse effects—including heightened agitation and anxiety—may restrict its initial usage. It is common practice to reserve fluvoxamine for cases in which the administration of sertraline or fluoxetine has proven ineffective(20).
The requirement for low-dose formulations of certain SSRIs may necessitate the engagement of a compounding pharmacist to prepare an appropriately concentrated liquid form, particularly for pediatric patients who may benefit from such adjustments. While sertraline, fluoxetine, and fluvoxamine have demonstrated efficacy in managing depressive disorders, there remains a notable dearth of robust empirical studies assessing their effectiveness specifically in children with ASD who concurrently present with mood disorders.
Consequently, treatment regimens should be developed on an individualized basis, following a thorough and specialized assessment (3). Moreover, it is imperative to acknowledge the associated risk of increased suicidal ideation linked to SSRI treatment, particularly during the initial phases of pharmacotherapy(11). Therefore, clinicians must meticulously evaluate the harm-benefit ratio specific to each patient, implementing rigorous monitoring protocols for any manifestations of suicidal thoughts during the first two weeks of treatment. Such measures are essential to ensure the safety and efficacy of therapeutic interventions within this vulnerable populations.
Mood lability: Children diagnosed with (ASD) who exhibit externalizing behaviors—defined as actions that are outwardly directed when experiencing distress, including physical aggression, verbal threats, and property destruction— commonly display a mood that is characterized by lability(21). This subset of children frequently confronts a range of challenges, including learning disabilities, difficulties with self-regulation, and notable behavioral issues within educational settings, which can impede their social and academic development.
Table 1: Drugs that can be considered for comorbidities in children with autism 14-17,27.
Pharmacological intervention may include the use of sodium valproate, a medication that has shown efficacy in mitigating aggressive behaviors while also addressing symptoms of irritability and mood instability commonly associated with ASD. Despite its clinical benefits, it is essential to recognize that current empirical evidence does not robustly support the use of other mood stabilizers specifically for this population. Sodium valproate is associated with a range of adverse effects, including, but not limited to, gastrointestinal disturbances such as nausea, cognitive impairments manifesting as poor attention, skin reactions such as Stevens-Johnson syndrome, and potential hepatotoxicity. Importantly, sodium valproate possesses significant teratogenic risks, particularly for females, and has been correlated with the development of polycystic ovary syndrome as well as Tourette syndrome. These factors necessitate a cautious approach to its prescription, particularly in vulnerable populations.
When managing tics and Tourette syndrome in these patients, clonidine is established as the first-line pharmacological treatment. Clonidine works primarily as an alpha-2 adrenergic agonist, which can effectively reduce tic frequency and severity. Additionally, there is a growing body of evidence supporting the use of atypical antipsychotics, such as aripiprazole, for this condition. Aripiprazole is known to facilitate symptomatic relief in Tourette syndrome and is considered particularly useful in cases where patients show inadequate responses to psychological interventions or where there are contraindications or insufficient efficacy associated with clonidine.
Furthermore, clinical observations have indicated that aripiprazole may be associated with a more favorable tolerance profile compared to clonidine, making it a compelling alternative for patients requiring pharmacological management for Tourette syndrome(22).
Therefore, the treatment of children with autism spectrum disorder and accompanying externalizing behaviors necessitates a meticulous evaluation of the risk-benefit profiles of available medications. This evaluation should be conducted in conjunction with a comprehensive assessment of the individual’s symptoms, comorbidities, and overall health status to ensure the most effective and safe treatment approach.
Irritability and aggression: Aggression presents a significant challenge for parents of children diagnosed with autism spectrum disorder (ASD), often leading to considerable disruptions in educational settings, interpersonal relationships, and overall familial cohesion.
The management of such behavioral issues is critical, as they can severely impede a child’s capacity to engage meaningfully in social and educational activities. Typically, pharmacological interventions have been employed to address these concerns, with antipsychotic medications such as aripiprazole and risperidone frequently utilized to ameliorate irritability and problematic behaviors in pediatric populations affected by ASD (24-25).
In the Australian context, risperidone has received approval from the Therapeutic Goods Administration for the treatment of irritability and aggression associated with autism in individuals under the age of 18. While risperidone can be effective, it is imperative to acknowledge its associated risks, which encompass weight gain, elevated levels of lipids, alterations in blood glucose, increased prolactin levels, and potential disruptions in normal puberty development. Conversely, aripiprazole, employed more widely in the United States, is noted for its favorable side effect profile, particularly in terms of weight gain and its lesser impact on prolactin levels, as well as its non-sedating properties. This characteristic can pose challenges for families who have relied on risperidone to manage nocturnal aggression and enhance sleep quality.
Clinical guidelines recommend a comprehensive assessment prior to the initiation of treatment with these antipsychotics. Essential baseline measurements should include height, weight, age of menarche, menstrual regularity, blood glucose levels, fasting lipid profiles, and prolactin levels. Subsequent monitoring is advised at one month and six months post-initiation of therapy. Should any elevations in prolactin or adverse motor manifestations occur, a reduction in dosage may be warranted, alongside a re-evaluation of the antipsychotic treatment regimen.
Current literature indicates that, despite the off-label use of other antipsychotic agents, including olanzapine, quetiapine, ziprasidone, and clozapine, there remains insufficient empirical evidence to substantiate their efficacy in managing aggression within the context of ASD, a finding supported by studies with limited sample sizes and non-randomized designs (10,11). Furthermore, older antipsychotic medications, antiepileptic drugs, and glutamatergic modulators—such as ketamine and memantine—exhibit little evidence of efficacy in mitigating aggressive behaviors (25).
In cases where standard antipsychotic treatments fail or are poorly tolerated, sodium valproate may be considered as an alternative therapeutic option. In select patients demonstrating inadequate response to risperidone or aripiprazole, the judicious off-label use of alternate antipsychotics may be contemplated, although such decisions must be made with thorough clinical consideration and monitoring (26).
Attention deficit hyperactivity disorder ADHD: Attention-deficit/hyperactivity disorder (ADHD) is often identified as a comorbid condition among individuals diagnosed with autism spectrum disorder (ASD), highlighting the need for a meticulous approach in both assessment and treatment (7,27).
The management of ADHD symptoms in individuals with ASD typically involves a spectrum of pharmacological interventions, which are categorized into stimulant and non-stimulant medications. Stimulant medications, including methylphenidate and dexamfetamine, have long been regarded as first-line treatments due to their robust efficacy in improving attentional focus and mitigating symptoms of hyperactivity and impulsivity. These agents function by increasing the availability of certain neurotransmitters, specifically dopamine and norepinephrine, in the brain, which facilitates enhanced concentration and behavioral regulation. Conversely, non-stimulant alternatives such as guanfacine, an alpha-2 adrenergic agonist, and atomoxetine, a selective norepinephrine reuptake inhibitor, offer viable options for patients who may not respond well to stimulants or who have coexisting anxiety disorders that may be exacerbated by stimulant use.
Atomoxetine, in particular, has gained recognition for its effectiveness in managing inattentive symptoms of ADHD, especially in the presence of anxiety, thus allowing for a more holistic management of the patient's condition (27). Clinical studies underscore the positive outcomes associated with the use of these pharmacological therapies, demonstrating significant improvements in ADHD-related symptoms among individuals with ASD. The enhancements in attentional capabilities and reductions in hyperactive-impulsive behaviors can contribute to improved academic performance, social interactions, and overall quality of life. However, practitioners must remain vigilant regarding the potential adverse effects that may accompany these treatments, including nausea, fatigue, and the possibility of sleep disturbances (28).
Therefore, developing individualized treatment plans that incorporate regular assessment of both therapeutic efficacy and side effects is paramount to achieving optimal outcomes in this multifaceted patient population. This comprehensive approach ensures that care is tailored to meet the unique needs of each individual, facilitating a better management framework for ADHD within the context of autism spectrum disorder.
Insomnia: Insomnia and sleep disorders are highly prevalent among individuals diagnosed with autism spectrum disorder (ASD), affecting approximately 80% of this population. These disorders primarily manifest as difficulties with sleep onset, which can significantly disrupt daily functioning and overall quality of life. The principal approach in managing these sleep-related issues is through behavioral interventions, which serve as the foundation for effective treatment.
Pharmacological options for addressing sleep disorders in children, especially those with ASD, are limited. The use of benzodiazepines is not recommended due to concerns regarding the potential for dependency and associated adverse effects. Similarly, antipsychotic medications are typically avoided due to their risk of significant negative side effects, which may outweigh any potential therapeutic benefits in this context.
Melatonin has gained recognition as a widely utilized adjunctive treatment for sleep disorders in children with ASD. Its availability as an over- the-counter supplement in numerous countries is notable, along with its low incidence of adverse effects and minimal risk of dependency.
Research indicates that melatonin may be particularly effective in facilitating sleep onset in this population. In addition, clonidine, when administered at low doses ranging from 50 to 200 micrograms at bedtime, has been identified as a potential treatment option, particularly when insomnia is secondary to stimulant medication used for attention-deficit/hyperactivity disorder (ADHD) (29). This approach should be incorporated into a comprehensive treatment strategy that emphasizes both behavioral modifications and tailored pharmacological interventions, ensuring that each child's care plan addresses their unique needs and circumstances (11).