¹Department of Chemistry and Industry, Faculty of Sciences, University of Kinshasa, P.O. Box 190 XI, D.R. Congo ²Department of Biology, Faculty of Sciences, University of Kinshasa, P.O. Box 190 XI, D.R. Congo
³Department of Biology and Applied Technology, Exact Sciences Section, Higher Institute of Education of Gombe, Kinshasa/Gombe, D.R. Congo
Abstract
The aim of this investigation was to assess the antioxidant and antiproliferative effects of the total methanol, dichloromethane, and aqueous extracts of Lannea antiscorbutica root bark, as well as to identify a few bioactive markers. The DPPH radical procedure was employed to assess the antioxidant activity. The methanolic, aqueous, and ascorbic acid extracts exhibit minimal antioxidant activity, with IC50 values of 1.72 μg/mL, 1.65 μg/mL, and 0.43 μg/mL, respectively. The dichloromethane extract produced a plateau that rendered it impossible to estimate the IC50. For 72 hours, the growth and germination inhibition of Sorghum bicolor seeds against the plant extracts were monitored to assess the antiproliferative activity. The lengths of all extracts, which ranged from 15.5 to 1000 µg/mL, were significantly shorter than the 32 mm blank. The dichloromethane extract yielded two steroids: Stigmast-4-en-3-one (1) and Stigmast-4-en-3,6-dione (2). The isolated compounds' structures were determined using 1H and 13C NMR, and the resulting data were compared to those in the literature. From these results, we concluded that this plant has antioxidant activity and can be fully applied for its activity against antioxidants and antiproliferative.
Keywords: Antiproliferative, DPPH, NMR, Stigmast-4-en-3-one (1) and Stigmast-4-en-3,6-dione (2), Lannea Antiscorbutica.
Introduction
The genus Lannea is a member of the Anacardiaceae family, which is primarily recognized for the production of polysaccharides, resins, or latex. Its leaves are alternate and lack stipulations (Van der Veken, 1960). About 40 species of trees, shrubs, and sub-shrubs with pinnate leaves, frequently adorned with star-shaped filaments, comprise the genus Lannea. In the Democratic Republic of the Congo, there are thirteen species and two varieties of these plants, which are found throughout tropical Africa and Asia (Avinash et al., 2011; Van der Veken, 1960). The isolation of a variety of secondary metabolites, such as alkylphenols, alkylhexenones, dihydroalkylhexenones, tetracyclic and pentacyclic triterpenes, tannins, phenolic lipids, benzoic acid derivatives, and flavonoids, has been the result of phytochemical studies conducted on the genus Lannea (Dorothy et al., 2014; Avinash et al., 2011; Emerson et al., 2003; Islam et al., 2002; Groweiss et al., 1997). The plant produces ovoid drupes that are green in color and appear prior to the foliage (Lathan, 2003; Van der Veken, 1960). The plant Lannea antiscorbutica is a versatile species that has significant traditional and medicinal applications. The bark is employed to treat anemia, typhoid fever, palpitations, pain, and tooth decay (Mukoko, 1991). The leaves are employed to alleviate symptoms of diseases such as diabetes, rheumatism, and diarrhea (Mukoko, 1991).
The decoction of pulverized bark of L. antiscorbutica, which is incorporated with the leaves of Carica papaya, is administered to alleviate toothaches, contracted muscles, and fractures, as well as to treat ophthalmia and orchitis (Mukoko, 1991). In the Province of Haut-Lomami in the Democratic Republic of the Congo, L. antiscorbutica is employed in ceremonies to invoke the spirits. It is a timber plant that is harvested for the purpose of constructing hedges (Göhre et al., 2016; Bakwaye et al., 2013; Lathan, 2004; Kambu, 1990). Polyphenols (tannins, flavonoids, anthocyanins), alkaloids, saponins, and terpenoids (diterpenoids and triterpenoids) are present in the species L. antiscorbutica, according to preliminary phytochemical investigations (Mvingu et al., 2017; Kabena et al., 2014).
The present study assessed the antioxidant and antiproliferative properties of the bark root of Lannea antiscorbutica using extracts. The dichloromethane extract of the root bark of L. antiscorbutica was used to isolate two known steroids: Stigmast-4-en-3-one (1) and Stigmast-4-en-3,6-dione (2). Thus, this species of the Lannea genus is the first to report the presence of these compounds. The objective of this investigation was to assess the antioxidant and antiproliferative properties of the root bark of L. antiscorbutica and to identify specific compounds.
Material And Methods
Plant Collection And Identification
The Lannea antiscorbutica root bark utilized in this investigation was obtained from Kisantu city, located in the Kongo-Central province of the Democratic Republic of the Congo. The plant was identified and verified by comparing it to the voucher specimen number H. Bruyne 2577 at the Herbarium of the National Institute of Agronomic Studies and Research (INERA) at the Faculty of Sciences at the University of Kinshasa.
Plant Extraction
The stem bark root of this plant was exposed to the elements for a period of one month. They were pulverized using an electric grinder (Blinder brand number B-592) and subsequently sieved. The substance that was then obtained was subsequently employed. Sorghum grains (Sorghum bicolor) were selected for this investigation in order to confirm their antiproliferative properties. These seedlings exhibit a rapid germination rate, which facilitates improved experiment performance.
The extraction was conducted using a sequence of solvents that increased in polarity: dichloromethane (DCM) was used initially, followed by methanol (ME), and ultimately distilled water (AQ).
- Three hundred (300g) grams of the plant powder were macerated in three liters of dichloromethane for 48 hours to produce the marc 1 (residue) after filtration and the filtrate, which, after evaporation, yields the dichloromethane extract;
- The same residual, marc 1, was macerated in two liters of methanol for 48 hours to produce pomace after filtration 2 and the filtrate, which, after distillation, yields the methanol extract;
- Pomace 2 was macerated in two liters of distilled water to produce pomace 3. The pomace was discarded, and the filtrate was used to produce the aqueous extract after distillation.
The filtering was performed using Whatman N°1 filter paper. Each solvent was evaporated using a Buchi Re 120 rotary evaporator under reduced pressure and dried in a Memmert oven at 40 °C. The concentrations that were obtained are extracts of dichloromethane extract (fraction 1), methanol extract (fraction 2), and water extract (fraction 3). The aqueous extract was used to assess the antiproliferative activity.
Antioxidant Activity
The antioxidant activity was assessed using 1,1-diphenyl-2-picrylhydrazyl (DPPH°) in accordance with the method described by Tailor and Goyal (2014) and Mohammad et al. (2011). A 900 μL volume of the freshly prepared methanolic solution of DPPH° (0.025 g/L) was supplemented with 100 μL of each extract at varying concentrations (1-6 µg/mL). In parallel, the blank control was generated by combining 100 μL of methanol with 900 μL of a methanolic solution of DPPH° at the same concentration as the original samples. The mixture was homogenized for 10 seconds using an agitator and incubated in a dark location at room temperature for 30 minutes. The absorbance was measured at 517 nm using a Uv-vis spectrophotometer (JENWAY 7315). The control was analyzed using the same procedure, and ascorbic acid was used as the standard.
The following formula was used to determine the percentage of inhibition of the DPPH radical:
With AC: absorbance of the control and AE: absorbance of the solution with the extract in different concentrations.
Evaluation Of Antiproliferative Activity
The antiproliferative activity of the extracts of the root bark of Lannea antiscorbutica on the seeds of Sorghum bicolor was determined using the method of Oise and Adisina, (2013), with a minor modification made. One thousand eight hundred seeds of Sorghum bicolor were cleansed and disinfected with ethanol for ten minutes, rinsed with distilled water, and subsequently desiccated in the open air. The seedlings' viability was assessed by their capacity to remain submerged in water for five minutes. Ayinde and Agbakwuru (2010) reported that all that remained submerged were recovered, sterilized by submerging in ethanol for 2 minutes, washed directly with water, and subsequently desiccated. Twenty-five milligrams of each extract were dissolved in a few droplets of 5% dimethylsulfoxide (DMSO) and subsequently diluted in 25 mL of distilled water. Concentrated solutions of varying dilutions were prepared in the following order: 1000 µg/mL, 500 µg/mL, 250 µg/mL, 125 µg/mL, 62.5 µg/mL, 31.3 µg/mL, and 15.6 µg/mL. A syringe was used to carefully transfer 4 mL of each extract concentration into 9 cm wide Petri dishes that had been previously filled with absorbent cotton containing twenty sterilized seeds. The plates are incubated in the shade and at room temperature for 72 hours. The number of germinated seeds in each container was enumerated after every 24 hours of incubation, and the rootlets were measured using a graduated slat. The control seedlings were only sprayed with 4 mL of distilled water devoid of plant extracts. The experiments were conducted in triplicate.
The subsequent formula was employed to determine the germination rate:
% germination
The antiproliferative activity was evaluated by the percentage of seed germination inhibition after three days and was calculated according to the following formula:
% inhibition germination = 100 - % germination
Separation And Purification Of Compounds
The Merck type silica gel 773429 of size ˂ at 0.06 mm was employed to isolate the compounds described in this investigation through column chromatography. The n-hexane-dichloromethane system was employed in varying proportions as the mobile phase. After separation and purification, TLC was performed on an F254 silica gel plate under ultraviolet light (at 254 and 366 nm) and/or a spraying agent composed of anisaldehyde, H2SO4, and methanol (1:2:97). The sample was then heated for a few minutes following the sprinkling.
Characterization And Identification Of Compounds
Interpretation of spectroscopic data (NMR) and comparison of chemical shifts in the literature were used to characterize and identify the compounds. All spectra were recorded at the ambient temperature with the Bruker 400 MHz spectrometer for 1H-NMR and the Bruker 125.76 MHz spectrometer for 13C-NMR in deuterated chloroform.
Results And Discussion
Yield And Chemical Screening
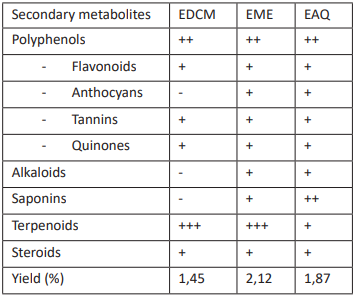
The extraction yield and the results of the chemical profiling conducted on the extracts are presented in Table 1. It is evident from this table that the methanolic extract (EME) (2.12%) yielded the highest yield, followed by the aqueous extract (EAQ) (1.87%), and the dichloromethan (EDCM) (1.45%) yield for Lannea antiscorbutica root bark. On the one hand, these variations may be attributed to the extraction technique and the character of the plant. Conversely, environmental factors that affect the quality and/or quantity of secondary metabolites of plants by affecting the expression of genes that regulate their synthesis (Falleh et al., 2008).
The presence of polyphenols (flavonoids, tannins, quinones, anthocyanins), terpenoids, and steroids in all extracts is indicated by the chemical screening Table 1. These secondary metabolites, which include anthocyanins, saponins, and alkaloids, are exclusively detected in polar extracts (EME and EAQ). These results are comparable to those of Dorothy (2014), Lannea barteri stem bark (Ibrahim, 2015), Lannea coromandalica bark (Vankatesan et al., 2015), and Lannea schweinfurthii (Kihagi, 2006). Lannea welwitschi has also been reported to contain saponins (Amiram et al., 1997). The presence of saponins in Lannea antiscorbutica may also facilitate the healing of micro lesions in the wall of a drained vagina that was caused by genital irritation during sexual intercourse (Kabena et al., 2014). The usage of this medicinal plant by African women for their intimate hygiene may be partially justified by the presence of these secondary metabolites, which could also mitigate the adverse effects of this practice, particularly the side effects (Kabena et al., 2014). The antiradical properties of phenolic compounds, including anthocyanins, flavonoids, and leucoanthocyanins, found in Lannea antiscorbutica, have the potential to inhibit or reduce the aging process of vaginal tissue (Ngbolua et al., 2014).
Legend: (-) negative test, (+) positive test and metabolite in small quantity, (++) positive test and metabolite in large quantity, (+++) positive test and metabolite in very large quantity.
Table 1: Yield and chemical screening of root bark extracts.
Antioxidant Activity
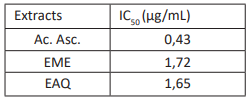
Table 2 indicates that the antioxidant activities of methanolic and aqueous extracts are lower than those of ascorbic acid (0.43 μg/mL): EME (1.72 μg/mL) and EAQ (1.65 μg/mL). It is important to mention that the dichloromethane extract (EDCM) exhibited a plateau, which rendered it impossible to estimate its IC50. The presence of phenolic and flavonoids compounds with antiradical properties was reported to be the reason for the elevated activity of the polar extracts (Hamidpour et al., 2015; Liu et al., 2009). In their investigation, Kafuti et al. (2018) determined that the IC50 value of ascorbic acid was 0.39 μg/mL, while that of methanolic extract and rutin was 1.99 μg/mL. These values are comparable to those of this investigation regarding polar extracts. Conversely, their hexanolic extract exhibited a high value (IC50 50.11 μg/mL), a result that was also observed in DCM extracts. Md. Arif Wahid (2009) conducted a study on Lannea coromandelica, which demonstrated antioxidant activity with IC50 values of 3.8, 6, and 8 μg/mL. Koné et al. (2011) found IC50 values of 4.71 and 3.54 μg/mL for EME of stem bark and roots in Lannea barteri, which were comparable to those of gallic acid (IC50 = 3.09 μg/mL) and ascorbic acid (IC50 = 3.14 μg/mL) when used as references. These values are distinct from those of ascorbic acid (0.43 μg/mL) and Lannea antiscorbutica extracts.
Anti-Proliferative Activity
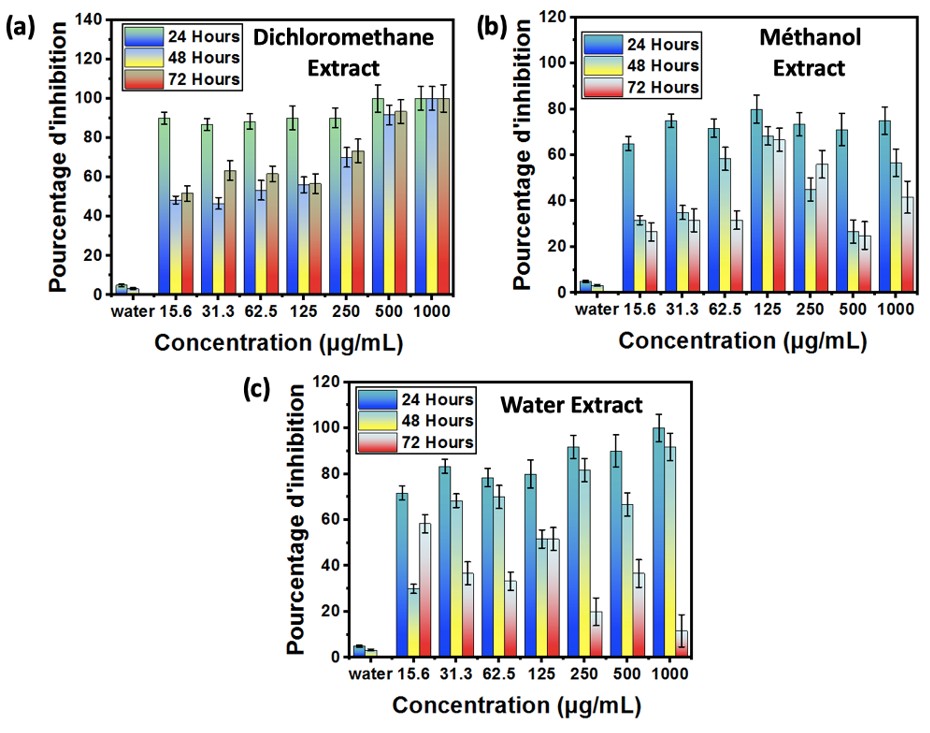
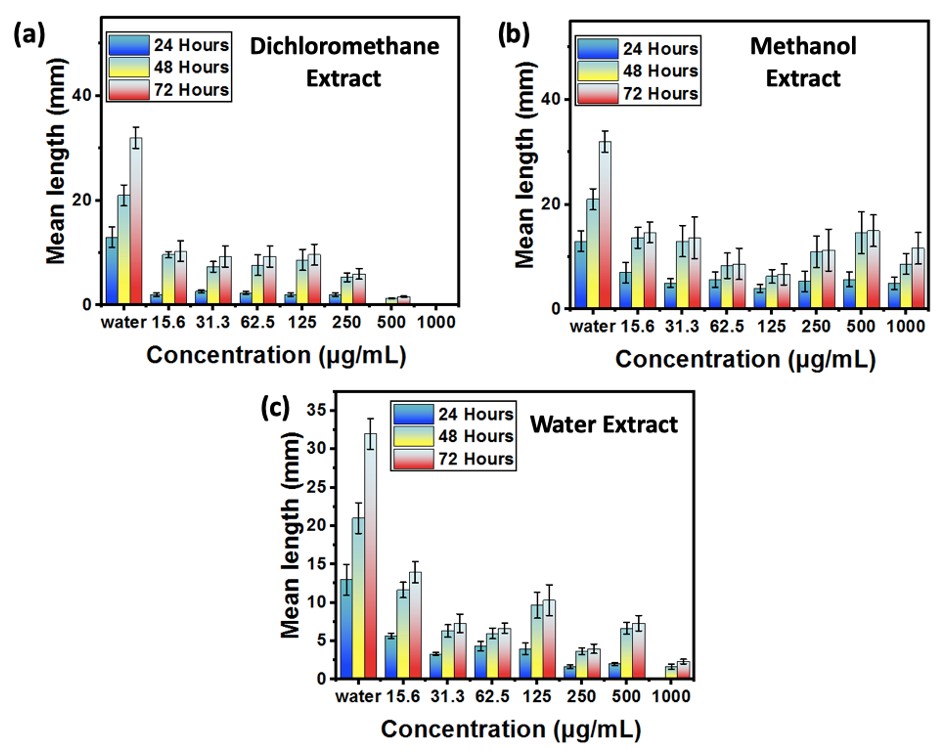
The antiproliferative activity was assessed by monitoring the germination and growth inhibition of Sorghum bicolor seeds against the plant extracts for 72 hours (Kafuti et al., 2018; Francis et al., 2018). The distilled water solution was employed as a negative control, and the values were also compared to those of Methotrexate MTX, an anticancer drug that was used as a positive control (Francis et al., 2018). The selection of the Sorghum bicolor as an in vitro model system is based on its simplicity, rapidity, reproducibility, and suitability for laboratories lacking cell lines (Ikpefan et al., 2013; Shobaike et al., 2002). The results of the effects of EDCM, EME, EAQ, and EAQD on the germination and growth of Sorghum bicolor seeds are presented in Figures 2 to 9.
Figure 2: Inhibition rate of dichloromethane, methanol and water extracts on Sorghum bicolor seed growth
Seed Germination Inhibition Rate
Fig. 2, (a, b and c) give the % germination inhibition of Sorghum bicolor seeds.
It is evident from these figures that seeds exhibit a percentage inhibition of 5%, 3.34%, and 0% at 24, 48, and 72 hours in the absence of treatment. This rate increased by the various concentrations of extracts employed following treatment. The DCM extract completely (100%) inhibited seed proliferation at 1000 µg/mL and by half (50%) at 15.6 µg/mL, in comparison to the DQAE (93.33% at 1000 µg/mL and 23.33%), EAQ (58.33% at 1000 µg/mL and 11.66% at 15.6 µg/mL), and EME (41.346% at 1000 µg/mL and 26.16% at 15.6 µg/mL), which are also higher than the control. The antiproliferative activity of methanolic extracts is verified by Francis Agada et al. (2018) in their research. In comparison to the extracts, the control exhibited seed proliferation (38 mm) at 72 hours. In contrast to the negative control, which exhibited a robust length growth of 93.77 mm at 72 hours, Abba and colleagues (2015) were able to neutralize this activity with the methanol extract. The antiproliferative activity of methanol and n-hexane extracts at 250 and 500 µg/mL was also substantiated by Kafuti et al. (2018) in their investigation of Boswellia dalzielii bark. The extracts exhibited high antiproliferative activity in the following order: EME (6.66±1.66 mm; 66.6%), EDCM (9.66±3.84 mm, 56.7%), and EAQ (10.33±0.33 mm; 51.66%) at the intermediate concentration (125 µg/mL). These findings corroborate the assertions made by the authors previously regarding the activity of polar versus nonpolar extracts. The rapid proliferation of cancer cells has been demonstrated in meristematic seed cells, such as those of Sorghum bicolor, when the conditions are favorable (Francis et al., 2018).
Micelle Length Of Seedlings
Fig. 3 (a, b and c) show the germination growth lengths of Sorghum bicolor seeds against white (the aqueous solution) and EDCM, EME, EAQ, EAQD.
These findings indicate that the lengths (mm) of all extracts at concentrations ranging from 1000 µg/mL to 15.5 µg/mL are significantly shorter than the blank length of 32.0±0.60 at 72 hours. Considering the two extreme concentrations, it was found that the DCM extract (Fig. 3a) showed no growth in 24 h at 500 µg/mL and in 72 h at 1000 µg/mL, a weak growth 1.33±0.33 (48 h), 1.16±0.33 (72 h) at 500 µg/mL compared to EAQ extracts (Fig. 3c) which showed no germination in 24 h at 1000 µg/mL, 1.66±0.66 (48 h), 2.33±0.88 (72 h) at 1000 µg/mL and 11.66±3.35 at 1000 µg/mL for EME (Fig. 3b) respectively. Based on the literature values of methotrexane, which are 0.60±0.01, 1.50±0.02, and 1.65±0.03 (Francis et al., 2018), it was observed that EDCM exhibits significant activity at 500 µg/mL and 1000 µg/mL, followed by EAQ.
Figure 3: Average germination growth length of Sorghum bicolor seeds of dichloromethane, methanol and water extracts
Characterization And Identification Of Compounds
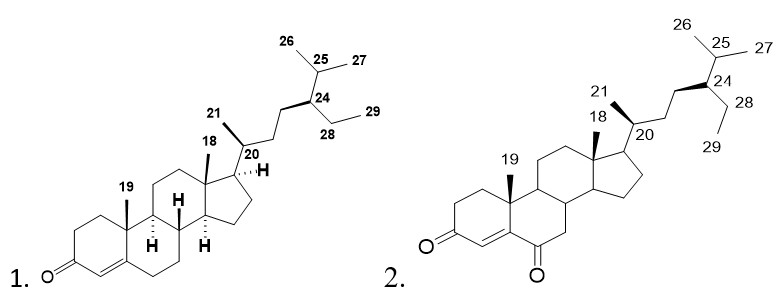
The first isolation of (24R)-Stigmast-4-en-3-one 1, (fig. 1), a white crystalline solid with the chemical formula C29H48O, was made in this species of the genus Lannea.
The 1H-NMR spectrum at 400 MHz, CDCl3 of this compound indicates six methyl group proton signals with chemical shifts from δ 0.71-1.18 ppm (s, 3H, CH3-19), which are indicated as follows: δ 1.18 ppm (s, 3H, CH3-19), 0. 92 ppm (t, 3H, J = 6.4 Hz, CH3-29), 0.82~0.84 ppm (9H, m, 3 CH3-21, 26, 27) and 0.71ppm (3H, s, CH3-18), a C-4 olefinic proton of the double bond at a weak field δ 5.72 (1H, s, H-4), δ 2.35 ppm (2H, m, H-2), δ 2.06 ppm (2H, m, H-6).
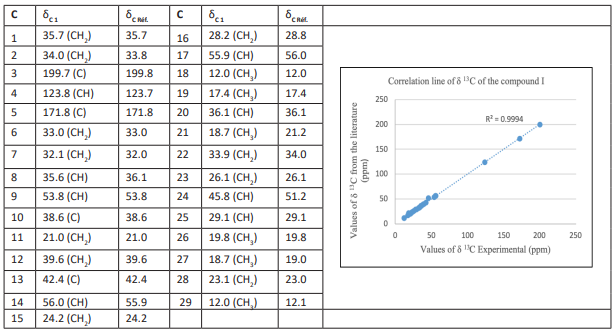
The 13C-NMR spectrum at 100 MHz, CDCl3 indicates signals of 29 carbon atoms in total, with one carbonyl carbon (C-3) signal at a weak field (δ 199.7 ppm) and two olefinic carbon signals at 123.8 ppm (C-4) and 171.8 ppm (C-5). It is also worth noting the presence of six methyl (C-18, 19, 21, 26, 27, 29), eleven methylene (C-1, 2, 6, 7, 11, 12, 15, 16, 22, 23, 28), eight methine (C-4, 9, 14, 17, 20, 24, 25) and four quaternary (C-3, 5, 10, 13) is also noteworthy. However, the literature data, when compared to these, indicate that compound 1 is a steroid that corresponds to (24R)-stigmast-4-en-3-one (Andi et al., 2017; Xiao et al., 2016; Kui et al., 2009). Recent research has demonstrated that this compound possesses a significantly higher α-glucosidase inhibitory activity than acarbose (IC50 127±0.64 mg/mL) and exhibits a very high anti-tumor activity (Habsah et al., 2005) (Nguyen et al., 2019). The 13C values for compound 1 are presented in Table 2 below:
The compound 2, Stigmast-4-ene-3,6-dione (fig. 1) with the chemical formula C29H46O2 was also isolated for the first time from Lannea antiscorbutica, and is present as a white crystalline solid.
The 1H-NMR spectrum at 500 MHz, CDCl3 indicates six methyl group proton signals with chemical shifts from δ 0.10-1.18 ppm corresponding to C including δ 1.14 (3H, s, CH3-19), 0.83 (t, 3H, J = 7.0 Hz, CH3-29), 0.78 ppm (3H, d, J = 6.8 Hz, CH3-27), 0.79 ppm (3H, d, J = 7. 0 Hz, CH3-26), 0.91 ppm (3H, d, J = 6.5 Hz, CH3-21) and 0.70 ppm (3H, s, CH3-18, a C-4 olefinic proton from the double bond to a weak field δ 6. 17 ppm (1H, s, H-4), δ 2.70, 2.06 ppm (2H, m, H-7), δ 2.54, 2.47 ppm (2H, m, H-2), δ 2.35 ppm (2H, m, H-1).
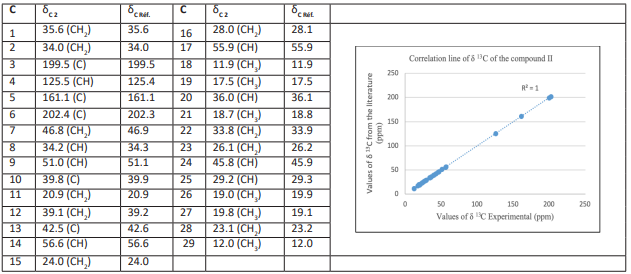
The 125 MHz 13C-NMR spectrum indicates signals from a total of 29 carbon atoms, with two low field carbonyl group signals (C-3, δ 199.5 ppm as in compound I and C-6, δ 202.4 ppm) and two olefinic carbon signals at 125.5 ppm (C-4) and 161.1 ppm (C-5). The presence of six methyl (C-18, 19, 21, 26, 27, 29), ten methylenes (C-1, 2, 7, 11, 12, 15, 16, 22, 23, 28), eight methines (C-4, 8, 9, 14, 17, 20, 24, 25) and five quaternaries (C-3, 5, 6, 10, 13) should also be noted. However, data from the literature compared to these show that compound II is a steroid corresponding to (24R)-stigmast-4-ene-3,6-dione (Kun et al., 2004; Tzong-Huei et al., 2005; Jae-Chul et al., 2005; Greca et al., 1990). The following biological activities have been reported: moderate antibacterial activity against gram (+) bacteria, Staphylococus aureus, Bacillus subtilis, Mycobacterum smegmatis (Hoai et al., 2017), moderate cytotoxicity against HL-60 and SMMC-772 cells (Wei-Yan et al., 2013), antigenotoxic activity against E. coli PQ37 (Jae-Chul et al., 2005) and moderate antitumor activity (Habsah et al., 2005). The 13C values of compound 2 are shown in Table 3 below:
Table 3: 13C values for compound 1, Ref. Andi et al., (2017)
Conclusion
Lannea antiscorbutica is a medicinal plant that is extensively employed in traditional Congolese medicine to treat a variety of ailments. A phytochemical screening was conducted on the fractions obtained by extraction with increasing polarity to validate its traditional use. The results of this study indicate that the highest yield was achieved in EME (2.12%), followed by EAQ (1.87%), and EDCM (1.45%). The extracts are abundant in polyphenols (including flavonoids, tannins, quinones, and anthocyanins), terpenoids, and steroids. Conversely, polar extracts (EME and EAQ) are the sole sources of alkaloids, saponins, and anthocyanins. These extracts exhibit a low level of antioxidant activity, as evidenced by their IC50 values of 1.72 μg/mL and 1.65 μg/mL, respectively, in comparison to ascorbic acid (0.43 μg/mL). There was no activity of the dichloromethane extract against the DPPH radical. The DCM extract completely (100%) inhibits seed proliferation at 1000 µg/mL and by half (50%) at 15.6 µg/mL, in comparison to EAQ (58.33% at 1000 µg/mL and 11.66% at 15.6 µg/mL), and EME (41.346% at 1000 µg/mL and 26.16% at 15.6 µg/mL), all of which are higher than the control. Two steroids, Stigmast-4-en-3-one (Compound 1) Stigmast-4-en-3,6-dione (Compound 2) was isolated from the dichloromethane extract and can be regarded as novel phytomarkers for chemotaxonomic investigations of Anacardiaceae. The utilization of the plant in traditional medicine in the Democratic Republic of Congo is partially supported by these findings.
Acknowledgment: Natural Products Research Group, Department of Chemistry, Faculty of Engineering and Physical Sciences, University of Surrey, Guildford, GU2 7XH, UK.
Declaration Of Competing Interest : The author declare that there is no competing financial interests in this paper