The Role of Fish Skin Xenografts in Healing Complex Wounds: A Brief Case Report
Camryn FDBS1, Naved BS1, Leslie SMSN2, Ahsan Raza MD,2
¹Research, Edward Via College of Osteopathic Medicine, Monroe, USA
²Department of Pharmaceutical Sciences, COMSATS Institute of Information Technology, Abbottabad 22060, Pakistan
*Corresponding author
*Camryn F. Daidone, Research, Edward Via College of Osteopathic Medicine, Monroe, USA
DOI: 10.55920/JCRMHS.2025.10.001423
Figure 1: CT scan taken during emergency department admission. The scan was remarkable for a large retroperitoneal abscess with lateral extension through the lower lateral abdominal wall musculature into the subcutaneous tissues. Area of retroperitoneal abscess posterior to the cecum and along the iliac is muscle measures approximately 15 cm in maximal length
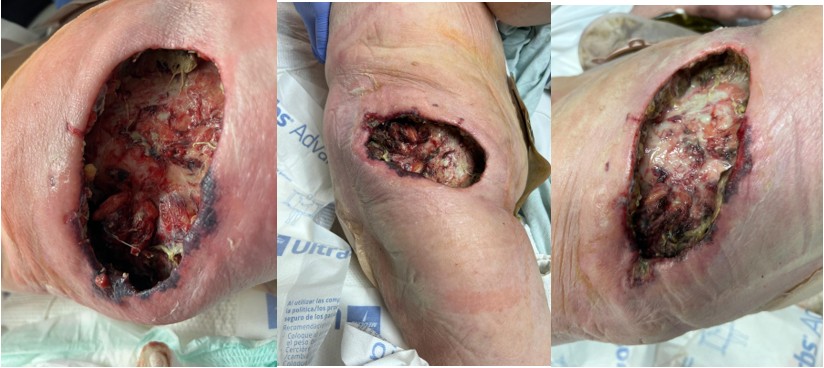
Figure 2 a-c: Right flank wound 2 days postoperative following wound debridement. The dimensions of this wound were 15cm by 10cm by 5cm deep.
Figure 3 a-c: Further debridement (3a), placement of xenograft (3b) and closure with wound VAC (3c). Dimensions of the wound were 17cm x 10cm x 4cm
Figure 4: Wound 5 days following xenograft placement. No signs of cellulitis, no tunneling, some sloughing of tissue
Figure 5: Wound approximately 1 week following xenograft placement. The wound appears healthy with granulation tissue present and no tunneling. At this point, the patient was discharged home with a wound VAC
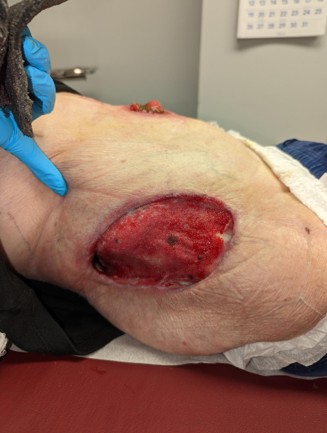
Figure 6: Wound on 28 day postoperative follow up visit, the wound is healed with substantial granulation tissue and the patient is scheduled for placement of a skin graft.
This patient was taken for an emergent exploratory laparotomy, drainage of abdominal and retroperitoneal abscess, open right hemicolectomy with diverting ileostomy, abdominal washout, intra-abdominal omental patch, placement of Strattice mesh for flank hernia prevention, and debridement of necrotizing soft tissue infection of right flank. Necrotic abdominal wall tissue and invasion of the hip joint was noted during surgery. There was stool contamination of the wound necessitating extensive debridement. The wound was 15cm x 10cm x 5cm deep and was initially debrided and washed with Dakins solution and placed on a Dakins wet-dry bandage. Figure 2 shows this wound 2 days after washing and debridement.
Two days post-operation, further debridement and application of a Kerecis™ xenograft with a wound Vacuum-Assisted Closure (VAC) was completed (Figure 3). The wound was followed over the next week in the hospital with periodic VAC changes as shown in figures 5 and 6 until the patient was discharged home and followed by home health for wound care. On postoperative day 28, a follow up visit revealed that the wound had healed well with substantial granulation tissue present. The patient was then scheduled for placement of a skin graft. The patient consented to use of clinical information and the included images in a case report and no personal health information was included to protect patient anonymity.