Decreased erythrocyte glyoxalase 1 (GLO1) activity in patients with diabetes with reduced estimated glomerular filtration rate
Rim Sakly1, Hiba Hamdi1, Amani Moussa1, Albert Lecube2, Hassen Bouzidi3, Baha Zantour4, Salwa Abid1, Mohsen Kerkeni1
1Laboratory of Research on Biologically Compatible Compounds, Faculty of Dental medicine, University of Monastir, Tunisia.
2Endocrinology and Nutrition Department, University Hospital Arnau de Vilanova. Obesity, Diabetes and Metabolism (ODIM) Research Group, IRBLleida. University of Lleida. Lleida, Spain
3Department of Biochemistry, CHU Tahar Sfar, Mahdia, Tunisia.
4Department of Endocrinology, CHU Tahar Sfar, Mahdia, Tunisia.
*Corresponding author
*Dr. Mohsen Kerkeni, Ph.D.; Higher Institute of Biotechnology, Avenue Tahar Haddad BP 74, 5000 Monastir,
Tunisia.
E-mail: mohsen.kerkeni@yahoo.fr
DOI: 10.55920/JCRMHS.2022.01.001024
Data are shown as the mean (SD) or median (range), or number (percentage).
NS: No significant
Biochemical parameters and GLO1 activity according the loss of renal function
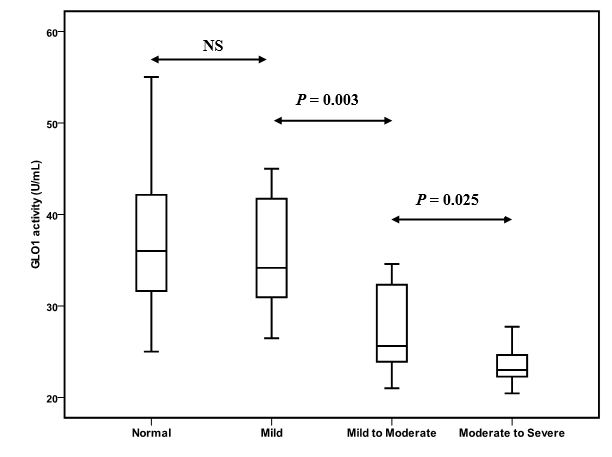
Clinical parameters and GLO1 activity in patients subgroups according eGFR were shown in Table 2 and Figure 1. Patients with diabetes were classified in four subgroup as normal, mild, mild to moderate, and, moderate to severe according eGFR. Duration of diabetes, glucose, and HbA1c did not differ between subgroups. As expected, eGFR was deceased from normal to severe subgroups (P < 0.001). For the GLO1 activity there was no difference between normal and mild group, however, a significant decrease was observed between mild to severe subgroups (P < 0.001).
Table 2: Biochemical parameters and GLO1 activity in diabetic patients subgroups according eGFR
Data are shown as the mean (SD) or median (range), or number (percentage).
**Significantly decreased between each group; P < 0.001
* Significantly decreased between Mild to severe group; P < 0.001
Figure 1: GLO1 activity in the subgroup diabetic patients according for the loss of renal function. (ANOVA analysis between the four subgroups: P < 0.001).
Correlation of GLO1 activity with eGFR and other variables
The GLO1 activity was correlated to eGFR (r = 0.257; P = 0.015) as shown in Figure 2. GLO1 activity was also correlated with serum creatinine (r= -0.328, p=0.002) and urea (r = - 0.300, P = 0.020,). Multivariate analysis showed that GLO1 activity was independently associated with eGFR (b = 0.129, P = 0.038). However, GLO1 activity did not shown any correlation with glucose, HbA1c, cholesterol, and triglyceride.
Figure 2: Correlation between GLO1 activity and the eGFR
- Kharroubi, A. T., & Darwish, H. M. Diabetes mellitus: The epidemic of the century. World journal of diabetes. 2015; 6(6), 850.
- Demakakos, P., Muniz-Terrera, G., & Nouwen, A. Type 2 diabetes, depressive symptoms, and trajectories of cognitive decline in a national sample of community-dwellers: A prospective cohort study. PLoS One. 2017;12(4), e0175827.
- Reece, E. A., Leguizamón, G., & Wiznitzer, A. Gestational diabetes: the need for a common ground. The Lancet. 2009; 373(9677), 1789-1797.
- Wu, Y., Ding, Y., Tanaka, Y., & Zhang, W. Risk factors contributing to type 2 diabetes and recent advances in the treatment and prevention. International journal of medical sciences. 2014; 11(11), 1185.
- Serván, P. R. Obesity and diabetes. Nutrición Hospitalaria. 2013; 28(5), 138-143.
- Kerkeni, M., Saïdi, A., Bouzidi, H., Yahya, S. B., & Hammami, M. Elevated serum levels of AGEs, sRAGE, and pentosidine in Tunisian patients with severity of diabetic retinopathy. Microvascular Research. 2012; 84(3), 378-383.
- Kerkeni, M., Saïdi, A., Bouzidi, H., Letaief, A., Ben Yahia, S., & Hammami, M. Pentosidine as a biomarker for microvascular complications in type 2 diabetic patients. Diabetes and Vascular Disease Research. 2013; 10(3), 239-245.
- Kerkeni, M., Weiss, I. S., Jaisson, S., Dandana, A., Addad, F., Gillery, P., & Hammami, M. Increased serum concentrations of pentosidine are related to the presence and severity of coronary artery disease. Thrombosis research. 2014; 134(3), 633-638.
- Knani, I., Bouzidi, H., Zrour, S., Bergaoui, N., Hammami, M., & Kerkeni, M. Increased serum concentrations of Nɛ-carboxymethyllysine are related to the presence and the severity of rheumatoid arthritis. Annals of clinical biochemistry. 2018; 55(4), 430-436.
- Knani, I., Bouzidi, H., Zrour, S., Bergaoui, N., Hammami, M., & Kerkeni, M. Methylglyoxal: A relevant marker of disease activity in patients with rheumatoid arthritis. Disease Markers. 2018.
- Vander Jagt DL, Robinson B, Taylor KK, Hunsaker LA. Reduction of trioses by NADPH-dependent aldo-keto reductases. Aldose reductase, methylglyoxal, and diabetic complications. J Biol Chem. 1992 Mar 5;267(7):4364-9.
- Thornalley, P. J. The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochemical Journal. 1990; 269(1), 1.
- Thornalley PJ. Glutathione-dependent detoxification of alpha-oxoaldehydes by the glyoxalase system: involvement in disease mechanisms and antiproliferative activity of glyoxalase I inhibitors Chem Biol Interact. 1998 Apr 24;111-112:137-51. DOI: 10.1016/s0009-2797(97)00157-9.
- Thornalley, P. J., Hooper, N. I., Jennings, P. E., Florkowski, C. M., Jones, A. F., Lunec, J., & Barnett, A. H. (1989). The human red blood cell glyoxalase system in diabetes mellitus. Diabetes research and clinical practice, 7(2), 115-120.
- Hamoudane, M., Amakran, A., Bakrim, N., & Nhiri, M. Decreased blood levels of glyoxalase I and diabetic complications. International Journal of Diabetes in Developing Countries. 2015; 35(3), 496-501.
- Pácal L, Chalásová K, Pleskačová A, Řehořová J, Tomandl J, Kaňková K. Deleterious Effect of Advanced CKD on Glyoxalase System Activity not Limited to Diabetes Aetiology. Int J Mol Sci. 2018 May 18;19(5):1517. doi: 10.3390/ijms19051517.
- Sakhi AK, Berg JP, Berg TJ. Glyoxalase 1 enzyme activity in erythrocytes and Ala111Glu polymorphism in type 1-diabetes patients. Scand J Clin Lab Invest. 2013 Mar;73(2):175-81. DOI: 10.3109/00365513.2013.765028. Epub 2013 Jan.
- Peters AS, Lercher M, Fleming TH, Nawroth PP, Bischoff MS, Dihlmann S, Böckler D, Hakimi M. Reduced glyoxalase 1 activity in carotid artery plaques of nondiabetic patients with increased hemoglobin A1c level. J Vasc Surg. 2016 Oct;64(4):990-4. DOI: 10.1016/j.jvs.2016.04.025. Epub 2016 Jul 29.
- Inagi R. RAGE and glyoxalase in kidney disease. Glycoconj J. 2016 Aug;33(4):619-26. doi: 10.1007/s10719-016-9689-8.
- Brouwers O, Niessen PM, Miyata T, Østergaard JA, Flyvbjerg A, Peutz-Kootstra CJ, Sieber J, Mundel PH, Brownlee M, Janssen BJ, De Mey JG, Stehouwer CD, Schalkwijk CG Glyoxalase-1 overexpression reduces endothelial dysfunction and attenuates early renal impairment in a rat model of diabetes. Diabetologia. 2014 Jan;57(1):224-35.
- Giacco F, Du X, D'Agati VD, Milne R, Sui G, Geoffrion M, Brownlee M.Knockdown of glyoxalase 1 mimics diabetic nephropathy in nondiabetic mice. Diabetes. 2014 Jan;63(1):291-9. doi: 10.2337/db13-0316.
- Schalkwijk CG, Stehouwer CDA. Methylglyoxal, a Highly Reactive Dicarbonyl Compound, in Diabetes, Its Vascular Complications, and Other Age-Related Diseases. Physiol Rev. 2020 Jan 1;100(1):407-461. DOI: 10.1152/physrev.00001.2019.
- Borg DJ, Forbes JM. Targeting advanced glycation with pharmaceutical agents: where are we now? Glycoconj J. 2016 Aug;33(4):653-70. DOI: 10.1007/s10719-016-9691-1. Epub 2016 Jul 9.
- Eringa EC, Serne EH, Meijer RI, Schalkwijk CG, Houben AJ, Stehouwer CD, Smulders YM, van Hinsbergh VW. Endothelial dysfunction in (pre)diabetes: characteristics, causative mechanisms and pathogenic role in type 2 diabetes. Rev Endocr Metab Disord. 2013 Mar;14(1):39-48. DOI: 10.1007/s11154-013-9239-7.
- Rabbani N, Xue M, Thornalley PJ. Methylglyoxal-induced dicarbonyl stress in aging and disease: first steps towards glyoxalase 1-based treatments. Clin Sci (Lond). 2016 Oct 1;130(19):1677-96. DOI: 10.1042/CS20160025.
- Schalkwijk CG, Miyata T. Early- and advanced non-enzymatic glycation in diabetic vascular complications: the search for therapeutics. Amino Acids. 2012 Apr;42(4):1193-204. DOI: 10.1007/s00726-010-0779-9. Epub 2010 Oct 20.
- Rabbani N, Xue M, Thornalley PJ. Dicarbonyl stress, protein glycation, and the unfolded protein response. Glycoconj J. 2021 Jun;38(3):331-340. DOI: 10.1007/s10719-021-09980-0. Epub 2021 Mar 1.
- Rabbani N, Thornalley PJ. Protein glycation - biomarkers of metabolic dysfunction and early-stage decline in health in the era of precision medicine. Redox Biol. 2021 Jun;42:101920. DOI: 10.1016/j.redox.2021.101920. Epub 2021 Feb 26.
- Rabbani N, Xue M, Weickert MO, Thornalley PJ. Reversal of Insulin Resistance in Overweight and Obese Subjects by trans-Resveratrol and Hesperetin Combination-Link to Dysglycemia, Blood Pressure, Dyslipidemia, and Low-Grade Inflammation. Nutrients. 2021 Jul 11;13(7):2374. DOI: 10.3390/nu13072374.