Emerging infectious agents: an unusual case of Metapneumovirus pneumonia in an adult patient
Author's: Bigoni T; Urbani F; Papi A; Greco GF*; Beccaria M
Respiratory Medicine, Respiratory Unit, Cardio-Thoraco-Vascular Department, Hospital ASST Mantua, Mantua, Italy
*Corresponding author
Graziana Francesca Greco, Medical doctor, Respiratory Unit, Mantua. E-mail: graziana.greco@asst-mantova.it
All Authors equally contributed to this manuscript
DOI: 10.55920/JCRMHS.2022.01.001001
A 61-yr-old caucasian man presented to the Emergency Department (ASST Mantua Hospital, Mantua, Italy) with fever up to 39°C, poorly responsive to antipyretics, nocturnal dyspnea and productive cough with mucus-purulent sputum for three days. On physical examination he appeared in good general condition, collaborating and oriented. The following parameters were recorded: blood pressure 140/90mmHg, heart rate of 100 beats min-1; respiratory rate of 23 breaths min-1; and body temperature of 38.4°C. His arterial oxygen saturation on room air was 87%. Chest examination revealed abnormal breath sounds with rhonchi and fine crackles in the middle lobe and inferior lobes bilaterally, no wheezes were heard. Laboratory findings revealed lymphocytosis (81000 x 103/µl), low platelet count (113000 x 106/µl) and an increase in alanine transaminase value (59 U/L), total bilirubin value (1.13 mg/dL) and CPR value (112 mg/L). Room air arterial blood gas analysis showed a normocapnic hypoxemia: pH 7.43, carbon dioxide tension 40.5 mmHg, oxygen tension 60.4 mmHg, and HCO3 24 mmol L-1. The SARS-CoV-2 antigen detection test on nasopharyngeal swab was negative. A chest radiograph showed multiple, small, patchy opacities in the right upper and middle lobe and no pleural effusion was observed. Based on these findings he was admitted to the Respiratory Department.
His medical history included chronic lymphocytic leukemia in follow-up which did not require any specific treatment. He denied taking any medications or to be a smoker, he drinks a glass of wine once a day and has no known allergies. The patient was a farmer who cultivates wheat and maize but he had no animal exposure and no travel history in the last few years. There is no family history or childhood history of respiratory complaints. He was vaccinated with three dosesagainst the SARS-CoV-2 infection (Pfizer) but not against the influenza virus.
Based on the patient’s presentation and testing results, on suspicion of bacterial pneumonia he was empirically treated with IV Piperacillin/Tazobactam, the patient required oxygen support at 3L min-1 and an inhalation therapy with Beclomethasone/Formoterol was set up ex adiuvantibus. In the following days, several microbiological investigations were carried out to determine the etiology of pneumonia: blood culture, urinoculture, sputum culture, Legionella, Haemofilus and Pneumococcus serologic tests, Legionella pneumophila and Pneumococcal urinary antigen test, all of which were negative.
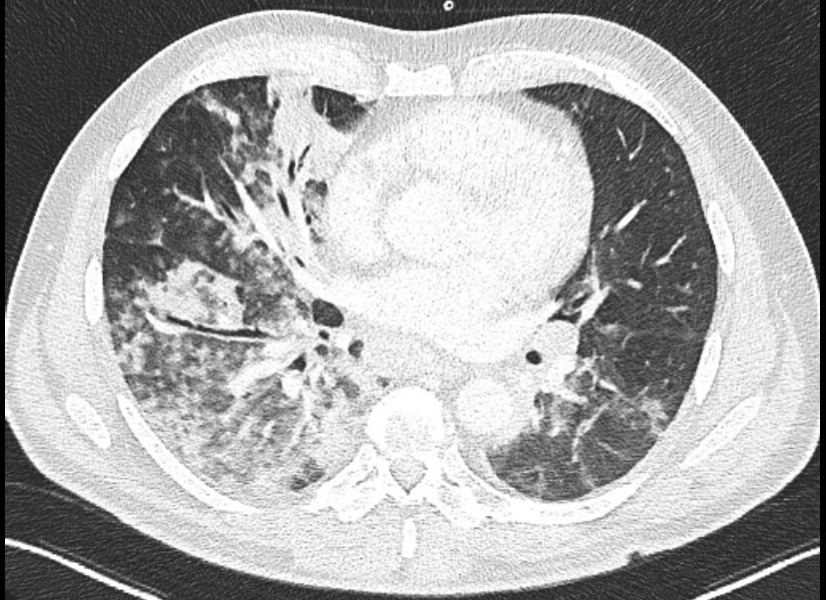
A nasopharyngeal swab FilmArray Respiratory Panel Assay (NP FARP) was then requested: it was positive for human Metapneumovirus and the result was confirmed by repeating the test. For non responder fever and further increase of CPR (230 mg/l) and PCT (0.27 ng/ml), Levofloxacin and later Meropenem were added in the perspective of a resistant bacterial etiology. On the 6th hospitalization day a chest computed tomography (CT) scan was obtained (Figures 1 and 2) which demonstrated large opacities with gradient borders, distributed in the peribronchial area at the right upper lobe, middle lobe and both the lower lobes; they tended to the confluence configuring parenchymal consolidations with aerial bronchogram at the level of the cost-phrenic angle. Imaging also showed bilateral hilar and mediastinal lymphadenopathy (max diameter 3.4 x 2 cm), splenomegaly and absence of pleural effusion. Blood chemistry tests for HIV, Aspergillus antigen and galactomannan were also investigated but turned out negative. To rule out other infectious agents the patient underwent bronchoscopy with bronchoalveolar lavage (BAL) into the middle lobe. BAL provides material for various microbiological and cytological tests: Gram stain, culture, Koch’s bacillus DNA, Galactomannan, Cytomegalovirus and P. Jirovecii and immunological analysis were negative. From respiratory virus panel on BAL only human Metapneumovirus was isolated, this unique microbiological data was according to the NP FARP’s result, thus supporting and confirming the new hypothesis of a viral pneumonia in an adult patient with probable secondary mild immunosuppression due to his hematological disease. About ten days after entering the ward, there was a gradual decrease of CPR and a progressive improvement in clinical conditions and respiratory function to allow the suspension of oxygen therapy. At the end of hospitalization, pulmonary function tests were performed and showed a restrictive syndrome (FEV1/FVC 76.2, TLC 68% and VC 79% of predicted) and mild reduction of diffusion capacity (DLCO 62% and KCO 99%), probably representing the residual functional impairment due to viral pneumonia. The patient finally suspended all therapies and at discharge was referred for a one-month follow-up visit.
Figure 1 : Horizontal slice of chest CT of a hMPV pneumonia
Figure 2 Chest computed tomography scan of a hMPV pneumonia demostrating parenchymal consolidations with aerial bronchogram
Human Metapneumovirus (hMPV), a relatively new virus first discovered in 2001, has been detected in 4-16% of patients with acute respiratory infections [1] [2] [3]. In particular, a recent review of 48 previous articles, including 100,151 patients under the age of five hospitalized for CAP, identified this virus as a cause of pneumonia in 3.9% of patients [4]. A recent study of 1386 hospitalized adult patients identified hMPV pneumonia in only 1.64%, indicating that it was much less common than in the infant population [5]. Metapneumovirus causes disease primarily in infants, but rarely can infect immunosuppressed individuals and elderly as well. Seroprevalence studies have shown that 90-100% of 5-10 years old children have previous infection [6]. Reinfection can occur during adulthood because of defected immunity acquired during the first contact with hMPV and/or because of different viral genotypes. The incubation period varies widely but is typically 3-5 days. The disease severity depends on the patient's condition and it ranges from mild upper airway infection to life-threatening pneumonia or bronchiolitis [7]. Clinically, Metapneumovirus infection is often indistinguishable from RSV infection, particularly in the pediatric population, and common symptoms include hypoxemia, cough, fever, upper and lower airway infections and wheezing [8]. hMPV infant patients are often hospitalized for bronchiolitis and pneumonia [9]. In young adults, a flu-like syndrome with fever may occur in a small number of instances, but infection in geriatric subjects may cause severe clinical manifestations such as pneumonia and, in rare cases, death [10].
As described in this case, it was not surprising that antibiotics and corticosteroids were administered in most patients infected with Metapneumovirus mainly for two reasons: in most cases the specific diagnostic tests for hMPV are not carried out at admission and/or physicians prefer to continue steroid and antibiotic treatment to control potential unidentified bacterial infections in patients in which no etiological agent had been identified associated with hMPV infection. The overuse of these drugs could therefore be reduced through the adoption at admission of specific diagnostic tests for such etiological agent, especially if specific risk factors are present (age, immunodepression, etc.). In addition, the adoption of such tests could reduce the nosocomial spread of this virus, allowing an early isolation of the infected patient [11].
Conflicts of interest: The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript. Funding: The authors report no involvement in the research by the sponsor that could have influenced the outcome of this work.
Authors’ contributions : All authors contributed equally to the manuscript and read and approved the final version of the manuscript.
- Turner P, Turner C, Watthanaworawit W, Carrara V, Cicelia N, Deglise C, et al. Respiratory virus surveillance in hospitalised pneumonia patients on the Thailand–Myanmar border. BMC Infect Dis 2013;13:434.
- Lu G, Li J, Xie Z, Liu C, Guo L, Vernet G, et al. Human metapneumovirus associated with community-acquired pneumonia in children in Beijing, China. J Med Virol 2013;85:138–43.
- Lo pez-Huertas MR, Casas I, Acosta-Herrera B, Garcı a ML, Coiras MT, Pe rezBren˜a P. Two RT-PCR based assays to detect human metapneumovirus in nasopharyngeal aspirates. J Virol Methods 2005;129:1–7.
- Ning G, Wang X, Wu D, Yin Z, Li Y, Wang H, Yang W. The etiology of community-acquired pneumonia among children under 5 years of age in mainland China, 2001-2015: A systematic review. Hum Vaccin Immunother. 2017 Nov 2;13(11):2742-2750.
- Walsh EE, Peterson DR, Falsey AR. Human metapneumovirus infections in adults: another piece of the puzzle. Arch Intern Med. 2008;168(22):2489-2496.
- Van den Hoogen BG, de Jong JC, Groen J, Kuiken T, de Groot R, Fouchier RA, et al. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat Med 2001;7:719–24.
- Panda S, Mohakud NK, Pena L, Kumar S. Human metapneumovirus: review of an important respiratory pathogen. Int J Infect Dis. 2014 Aug;25:45-52.
- Boivin G, De Serres G, Cote S, Gilca R, Abed Y, Rochette L, et al. Human metapneumovirus infections in hospitalized children. Emerg Infect Dis 2003;9:634–40.
- Matsuda S, Nakamura M, Hirano E, Kiyota N, Omura T, Suzuki Y, et al. Characteristics of human metapneumovirus infection prevailing in hospital wards housing patients with severe disabilities. Jpn J Infect Dis 2013;66:195– 200.
- Falsey AR, Erdman D, Anderson LJ, Walsh EE. Human metapneumovirus infections in young and elderly adults. J Infect Dis 2003;187:785–90.
- Van den Hoogen BG, van Doornum GJ, Fockens JC, Cornelissen JJ, Beyer WE, de Groot R, Osterhaus AD, Fouchier RA. Prevalence and clinical symptoms of human metapneumovirus infection in hospitalized patients. J Infect Dis. 2003 Nov 15;188(10):1571-7.